Epoxy Joins Implantable Devices—and Protects Patients

One of the most challenging applications for an epoxy compound is bonding a fully implantable medical device. Not only must the epoxy withstand sterilization procedures, but it must also exhibit biocompatibility for an extended period of time. Both capabilities are essential for the device to maintain high performance over the long term—even when in direct contact with tissue and bodily fluids—and prevent the introduction of toxins that can cause injury, a physiological reaction or immunological rejection.
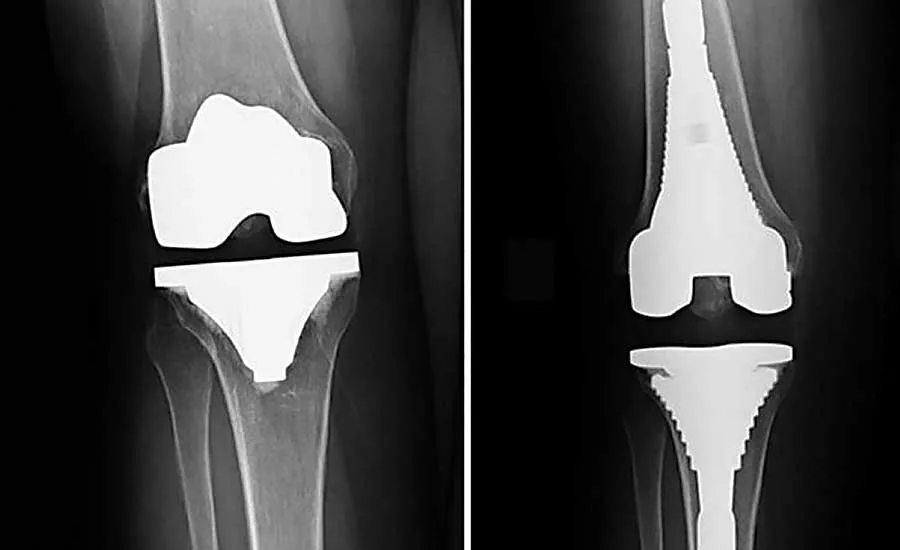
A prosthetic knee is a common implantable device that consists of three major parts: a tibial component, which is connected to the shinbone; a femoral component, which is connected to the thighbone; and a meniscal-bearing component (or spacer), which is located between the other two components and allows them to slide over each other.
The tibial and femoral components are usually made of metal or a metal alloy, while the bearing component is made of a plastic, such as polyethylene. Early designs of prosthetic knees fixed the bearing component to the tibial component, but newer designs allow the bearing component to float to some extent, allowing increased freedom of movement within the knee. However, all knee prostheses incur some risk of dislocation or spinout, especially if the ligaments fail to provide adequate support. Ideally, a prosthetic knee should allow for some flexion and tension of the knee joint, while lowering the risk of dislocation and bearing spinout.
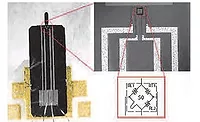
In 2014, an inventor at Howmedica Osteonics Corp. in Mahwah, NJ, was awarded a patent for his design of a limited motion tibial implant. The design calls for the addition of an elastic gasket between the tibial component and the meniscal-bearing component of a prosthetic knee.
Within the patent, the two-component EP42HT-2Med epoxy from Master Bond Inc. is cited as a suitable adhesive for mounting the gasket on a plate. This gasket-plate combination is designed to fit (without affixing) into an undercut rim on the top surface, or baseplate, of the tibial component. A lip around the top perimeter of the gasket is designed to couple with a groove in the bearing component.
When the three parts are sandwiched together, the gasket fills the space between the inner wall of the tibial component and the outer wall of the bearing component. The bearing component is free to move along the tibial baseplate, and as it moves, it compresses the gasket. However, the constant spring and thickness of the gasket restrict the bearing-component movement along the tibial baseplate. Thus, the design provides for limited motion with enhanced stability, unlike designs with floating bearing components.
The inventor selected the EP42HT-2Med epoxy for several reasons. Most importantly, the epoxy can withstand repeated sterilizations, including ethylene oxide processing, radiation, chemical sterilants, and autoclaving. It offers excellent bonding, sealing and coating, and is castable to thicknesses of 2 to 3 inches.
Looking for quick answers on assembly and manufacturing topics? Try Ask ASM, our new smart AI search tool. Ask ASM
In addition, the epoxy is highly resistant to many acids, bases, solvents, and fuels; offers excellent electrical insulation properties; and is serviceable from -60 to 450 F. It cures at room temperature or more rapidly at elevated temperatures. However, optimal performance and biocompatibility can be achieved by curing overnight at room temperature and then for an additional 2 to 4 hours at 150 to 200 F.
Available in both amber-clear and black as a USP Class VI system, the EP42HT-2Med epoxy is often used in the assembly of other medical devices, as well as surgical instruments, catheters and endoscopes. For more information, call 201-343-8983 or visit www.masterbond.com.
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!