Welding Incompatible Thermoplastics
Incompatible thermoplastic polymers can be welded by using polymer blends that are compatible with both components.
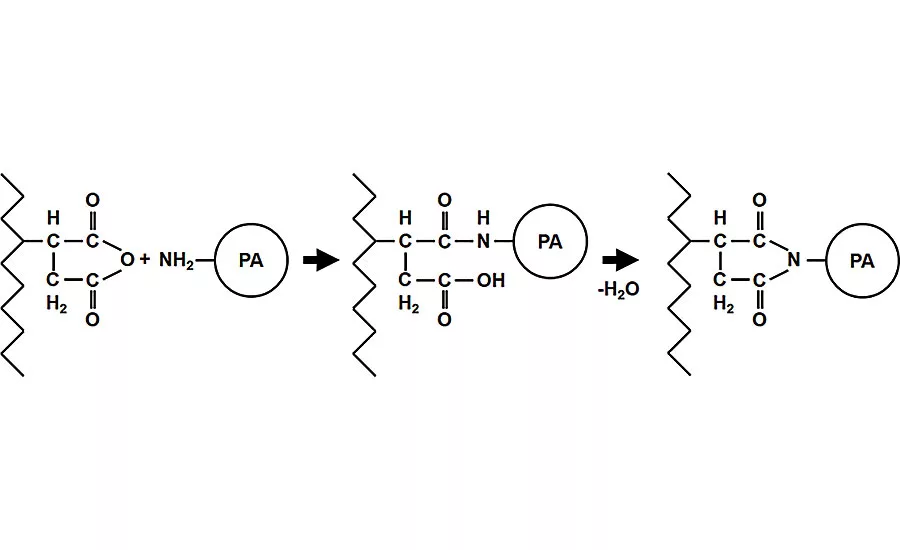
The maleic anhydride groups are the most commonly used coupling agents. The maleic anhydride is grafted on the PE and the pendent anhydride groups then react with the polyamide amine end-groups. This illustration shows the reaction of a succinic anhydride group in the modifie
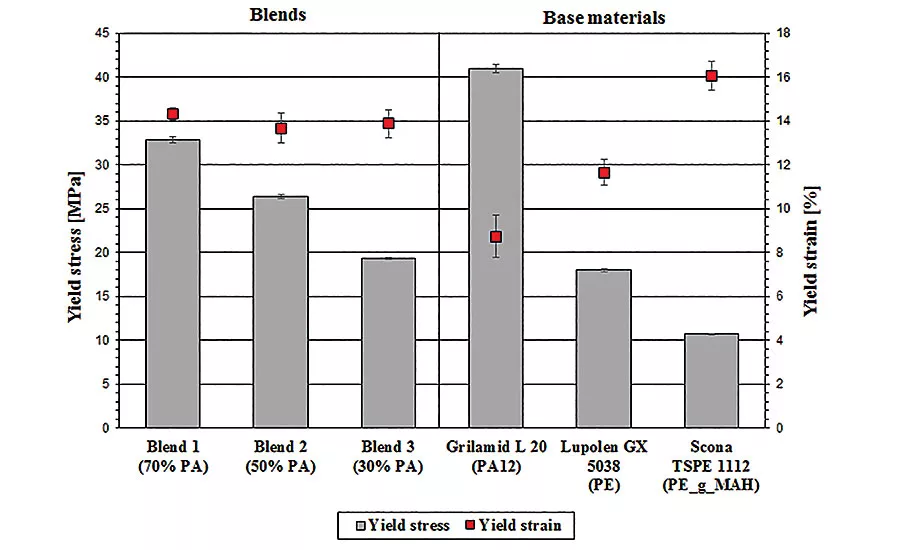
This graph summarizes the results for yield stress and strain. Values for the base materials are plotted on the right to serve as a comparison. The yield stress of the polymer blend decreases with increasing HDPE content, while the yield strain remains constant at around 14 percent and is higher than that of the base materials.
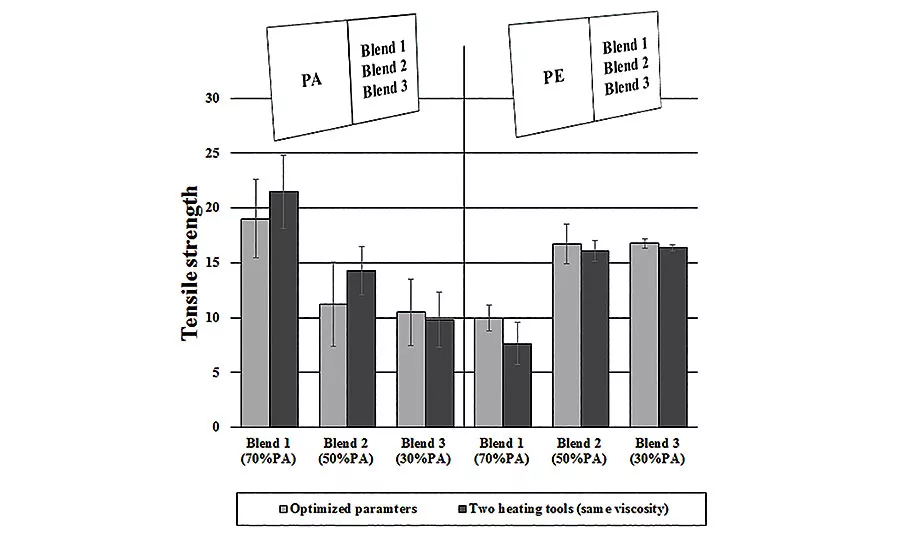
This graph illustrates the tensile strength achieved when the authors used hot-plate welding to join their polymer blends to HDPE and PA12. The gray bars show the results obtained with their original optimized settings. The dark bars show the results when they used two separate heating tools to tailor the heat to each part.
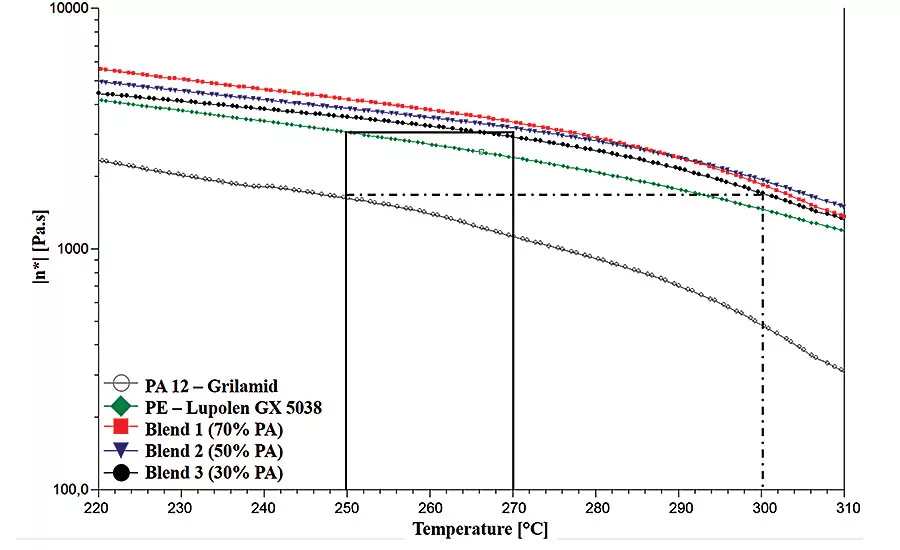
This graph illustrates the melt viscosity of the polymer blends and base materials at varying temperatures. The viscosity of the blends is higher than the viscosity of the base materials, due to the higher molar mass of the blends.

The authors used hot-plate welding to join a PA12 part to a coupon made from a blend of 70 percent PA12 and 30 percent HDPE. Surprisingly, the welding seam contained several small micro-cracks.
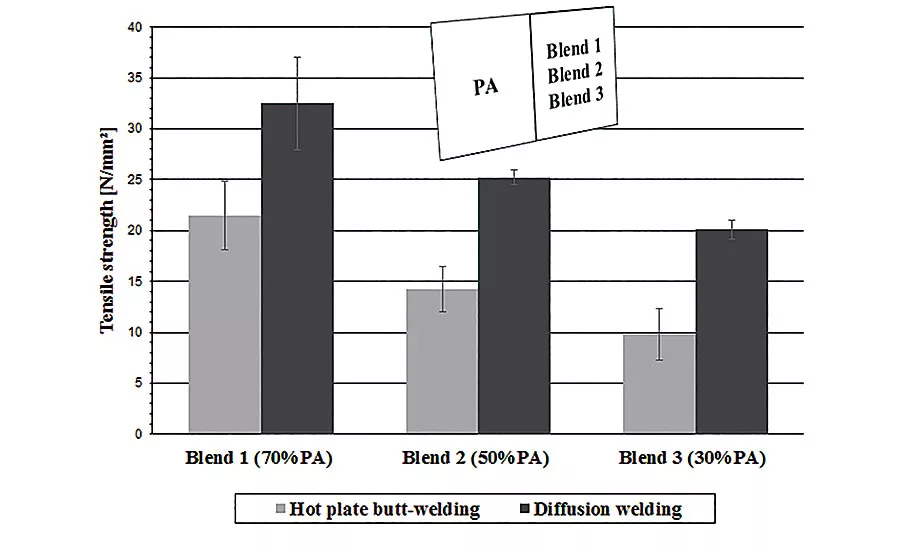
This graph compares the tensile strength achieved with hot-plate welding (grey bars) and diffusion welding (dark bars).

This photo shows fracture patterns after tensile testing of parts joined with diffusion welding. For joints with blend 1 (70 percent PA12), the fracture path goes through the base material, and the fracture starts in the most fragile polymer part—the blend.

Ideally, every component in a plastic assembly would be made from the same material. However, cost, functional and aesthetic requirements often dictate that a mix of materials be used.
Unfortunately, such design requirements are not always in synch with assembly requirements. As a result, engineers often have difficulty welding dissimilar plastics, due to chemical incompatibility or widely different melt temperatures.
To solve that problem, we set out to demonstrate the weldability of two incompatible thermoplastic polymers by using their blends. We learned that two incompatible thermoplastic polymers could be welded by using polymer blends that are compatible with both components. We chose two industrially relevant thermoplastics—high-density polyethylene (HDPE) and polyamide 12 (PA12)—to demonstrate the potential of an innovative joining technology.
The Blends
Ever since plastics were first discovered in the 19th century, chemists have been blending various polymers to achieve specific properties.
There are two types of blends: miscible and immiscible. While each polymer in an immiscible blend has its own glass-transition temperature (Tg), miscible blends have only a single combined one.
Most polymer blends are immiscible. In the absence of a coupling agent, polymer blends typically show no improvement of their properties over those of their individual components. Among the several types of coupling agents available, the maleic anhydride groups are the most common. The maleic anhydride is grafted to the polyethylene, and the pendent anhydride groups then react with the polyamide amine end-groups.
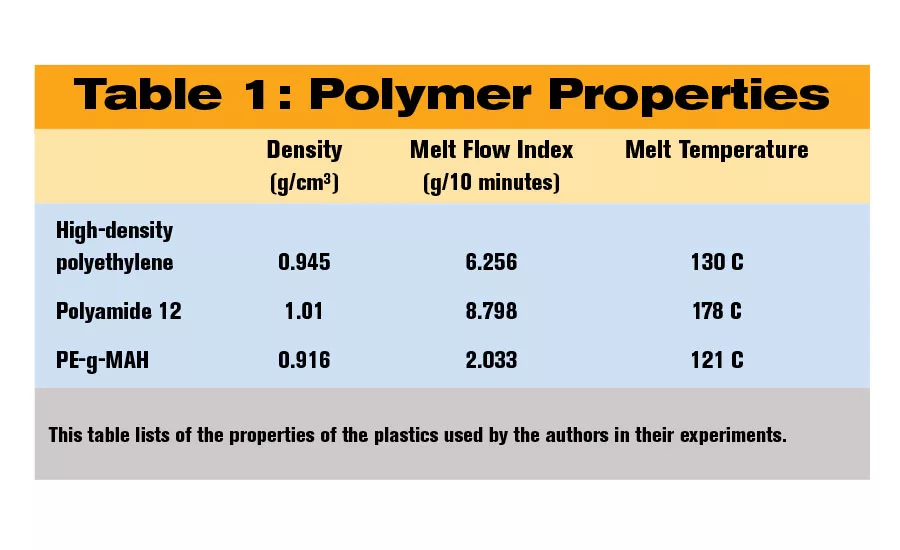
All the materials in our study were industrial grade. For the HDPE, we used Lupolen GX 5038 from Lyondell Basell. For the PA12, we used Grilamid L 20 from EMS-Grivory. For a compatibilizer, we used SCONA TSPE 1112 GALL from BYK, a linear low-density polyethylene that has been grafted with 2 percent maleic anhydride (PE-g-MAH). Also referred to as coupling agents, compatibilizers are additives that, when added to a blend of immiscible materials during extrusion, modify their interfacial properties and stabilize the melt blend.
Looking for quick answers on assembly and manufacturing topics? Try Ask ASM, our new smart AI search tool. Ask ASM
To eliminate the effects of moisture, the materials were dried for six hours at 85 C before blending. The blends of PA12, PE-g-MAH and HDPE were mixed using a co-rotating, twin-screw extruder. The blends were prepared in the following weight ratios:
Blend 1—70 percent PA12, 30 percent HDPE.
Blend 2—50 percent PA12, 50 percent HDPE.
Blend 3—30 percent PA12, 70 percent HDPE.
The compatibilizer represented 25 percent of the weight of the HDPE.
The test specimens (150 millimeters long, 75 millimeters wide and 4 millimeters thick) were injection molded.
The Welding Processes
For our study, we looked at hot-plate welding and diffusion welding.
The hot plate welding is a well-understood process, but the literature on welding of dissimilar polymers is quite small. Most studies are concerned with the welding of different grades of HDPE or polypropylene homopolymer to a polypropylene copolymer. However, the weldability of dissimilar polymers has not been researched systematically.
Hot-plate welding is one of the most widely used methods for joining plastics because of its great process stability. The process has four main phases. In phase 1, the part’s surface is brought into contact with the heating tool’s surface until the molten plastic begins to flow out laterally to ensure a full contact area.
In phase 2, the pressure is reduced so that the plastic part can be heated without application of additional pressure. This allows the molten film to increase in thickness homogeneously.
When a defined molten film thickness is created, the part and tool are separated in phase 3. At this stage, great care must be taken to avoid adhesion of the plastic to the heating tool. Furthermore, the time between heating and welding should be minimized, so the molten plastic doesn’t cool prematurely.
In phase 4, the molten surfaces of the two parts are brought into contact and pressed against each other under a predefined pressure until the interface solidifies. During this phase, the molten plastic flows laterally while cooling down.
An alternative welding process without a melt flow is diffusion welding. Like hot-plate welding, a tool is used to heat the joint interface to the melting point of the plastic. But, unlike hot-plate welding, the parts are assembled prior to heating, and no additional pressure is applied. This diminishes any undesired deformation in the joining plane. During the melt phase, macromolecules from each part can pass the joining plane until the heating tool is switched off, and the plastic solidifies as it cools. The bond is established by interdiffusion of the polymers.
Because of its long process time, diffusion welding is mostly considered irrelevant for industrial purposes. However, it is a good process with which to analyze the specifics of our system for joining dissimilar plastics.
Welding Experiments
For the hot-plate welding experiments, we initially used one tool, heated to a temperature that would accommodate the different melt temperatures of the plastic components. The temperature of the tool was between 220 C and 300 C. Welding pressure varied from 0.1 to 2 megapascals (MPa).
First, the base materials were welded without blends to characterize their maximal weld strength. Then, the polymer blends were welded with each other. Finally, the base materials were bonded by mediation of the polymer blends.
All process parameters were optimized prior to the start of the welding tests.
For the diffusion welding experiments, the test specimens were cut and milled to a size of 70 by 70 millimeters. They were inserted into a mold to prevent leakage of the melt under the subsequent applications of a preload force, which was needed to guarantee contact between the molten plastic. As a result of pilot tests, a heating tool temperature of 200 C and a heating time of 1 hour were chosen.
After welding, the test plates were cut into specimens for quasi-static tensile testing. The test plates that were joined with hot-plate welding were cut into eight test specimens to analyze their strength. The test plates that were joined with diffusion welding were cut into four test specimens. Both outer test specimens were not analyzed to eliminate marginal influences. We used a tensile testing machine with a 10 kilonewton load cell. The cross-head speed was 20 millimeters per minute.
Results and Discussion
Graph 1 summarizes the results for yield stress and strain. Values for the base materials are plotted on the right to serve as a comparison. The yield stress of the polymer blend decreases with increasing HDPE content, while the yield strain remains constant at around 14 percent and is higher than that of the base materials.
Yield stress and strain were found to be rather sensitive to force transmission between phases. According to this, yield stress and strain can be regarded as
suitable measures to evaluate the adhesion between different phases. A smaller
domain size shows a better dispersion of the polymers and, consequently, a better interfacial adhesion.
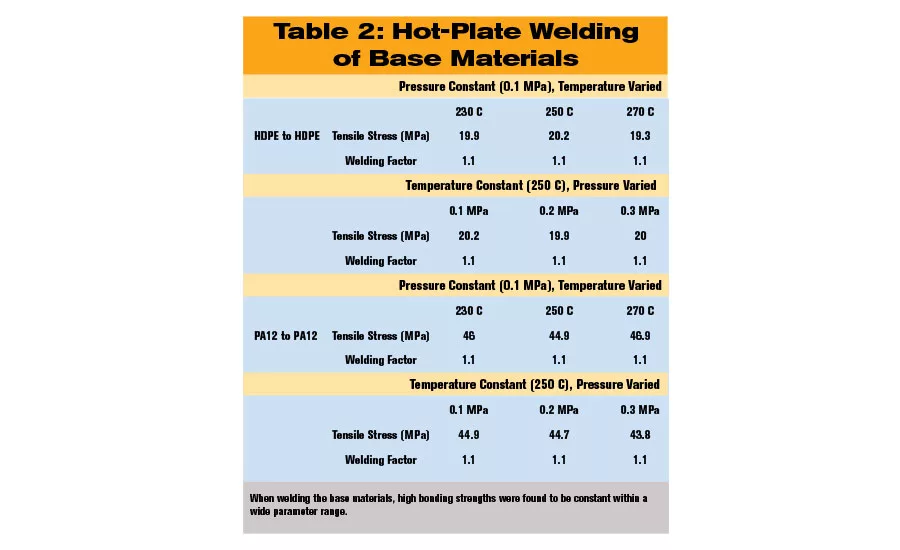
The welding experiments of the base materials show for both, polyethylene and polyamide, a welding factor of 1.1, which is illustrated in Table 2. (The welding factor, fz, is defined as the quotient of the weld strength, σWS, and the yield stress of the base materials, σBM: fz = σWS / σBM.)
These high bonding strength values are found to be almost constant within a wide parameter range for heating tool temperature and welding pressure. The variation in pressure is exemplarily shown for the temperature of 250 C. The welding factors greater than 1 were caused by the increase of the cross-sectional area of the weld bead.
Welding tests on the polymer blends were always done with blends of the same weight ratio. The welding factors of these experiments are shown in Table 3. The results show that a decreasing ratio of PA12 causes an increase of the welding factor. A reason for this could be the dominant phase of the polymer blend. Up to a ratio of 50 percent of PA12, HDPE seems to be the dominant phase. As long as HDPE is the dominant phase, it can be seen that a slightly higher bonding strength (around 19 MPa) can be achieved in comparison to the tensile strength of pure PE (18 MPa). The welding factor decreases only because of the higher tensile strength of the polymer blend.
Surprisingly, if HDPE is the minor phase in the blend, the weld strength decreases. However, the weld strength should be higher once PA12 is the dominate phase, because of the higher tensile strength of pure PA12. The theory of the weld formation mechanism is based on the relaxation of the molecular chains and the diffusion through the joining plane. As long as PE is the dominate phase, this theory can be considered valid.
If PA12 is the dominate phase, the diffusion process seems to be restricted. The reason for that could be the HDPE, which hinds the diffusion of the PA12 chains through the joining plane.
Furthermore, the mobility of the polymer chains is important for the diffusion process. The mobility of the PA12 chain is restricted because of the chemical bonding of PE-g-MAH. Due to the restricted mobility, the diffusion process decreases. Also, the unmodified side of the PA12 chain is only affine to other PA12 chains. For that reason, just this part of the chain can generate entanglements with other PA12 chains.
On the contrary, HDPE is affine to HDPE chains and modified PA12 chains. This supports the diffusion through the joining plane, so the weld formation mechanism.
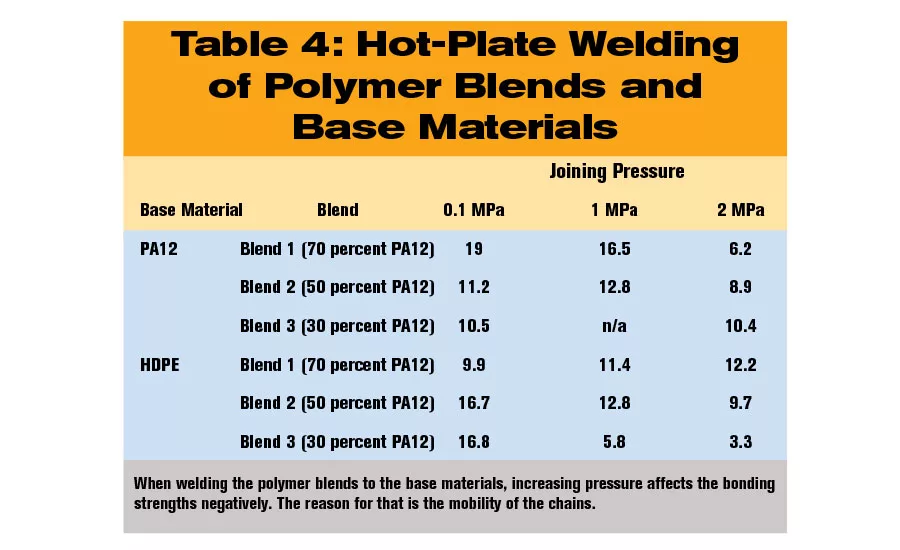
Welding Blends to Base Materials
The welding experiments of blends to base materials were done with the optimized parameters for the base materials. Thus, the heating tool temperature was 230 C for welding a blend to HDPE and 270 C for welding a blend to PA12.
As might be expected, the highest weld strengths are achieved with a high ratio of the base material in the blend. It is noticeable that the bonding strength of blend 2 (50 percent PA12) to HDPE already reached the level of the tensile strength of HDPE (18 MPa). On the contrary, the welding of blend 1 (70 percent PA12) to PA12 did not nearly reach the tensile strength level of the polymer blend. Here the comparison with the polymer blend instead of PA12 is required, because the blend has the lowest tensile strength of all involved components. The results confirm the expectation of the restricted mobility of the PA12 chains.
Considering the welding pressure, it can be seen that increasing pressure affects the bonding strengths negatively. The reason for that is the mobility of the chains. The molten layers on each plastic part possess temperature gradients. The temperature of the molten plastic is highest at the heating tool. But, with increasing distance from the tool, the temperature decreases. Increasing the welding pressure pushes the hottest molten material out of the welding zone, leaving somewhat cooler material to flow into the joining plane. With lower temperatures, the mobility of the molecular chains decreases and the diffusion through the joining plane is aggravated.
Due to the restricted chain mobility, we realized that we had to modify the welding parameters so the molten plastic of each part was at the same viscosity. This required us to heat both components to two different temperatures, which we achieved by using two heating tools.
To see if our thinking was correct, the polymer blends and base materials were analyzed rheologically. Graph 3 illustrates the melt viscosity of the polymer blends and base materials at varying temperatures. It can be observed that the viscosity of the blends is higher than the viscosity of the base materials, which is caused by the higher molar mass of the blends.
Following up, the tool for HDPE and PA12 was heated to temperature of 250 C, while the tool for the blends was heated to 270 C or 300 C, depending on the ratio of HDPE to PA12. If the weld strength is taken into account, the results, illustrated in Graph 2 (dark bars), show on one hand, a small increase for the welding of blends to PA12. On the other hand, the joint strength for the welding of blends to HDPE decreases. This could be a consequence of the increased chain mobility of PA12 within the blend that results in a better diffusion. However, this increased mobility could restrict the movement of the HDPE chains, which, in turn, would explain the slight drop in the tensile strength for the weld of blend to HDPE.
In this experiment, higher weld strengths for the welding of the blend to PA12 were expected. As the blend is always the weakest component, the weld strength should be at one level with the respective tensile strength. In contrast to the welding of blend 3 to HDPE, having a welding factor of 0.93, the welding of blend 1 to PA12 shows a welding factor of only 0.65.
The reason for that decrease surprised us. In the vicinity of the PA12-blend interface, solely on the part of the blend, the welding seam contained several small micro-cracks which emerged in the flow channel lengthways to the orientation of the molecular chains. We do not yet have a plausible explanation for the formation of those cracks.
To eliminate the flow process of the melt, diffusion welding experiments were performed. In these trials, the polymer blends were welded to PA12. Graph 4 shows a weld strength plateau at the level of the tensile strength of the involved polymer blend, which indicates a welding factor of nearly 1. This particular joint is corroborated by the absence of micro-cracks. It can therefore be assumed that the flow of the melt influences the polymer structure, so that the interfacial adhesion decreases. It is worth highlighting that, despite the low ratio of PA12, the welding
factor is still nearly 1.
For joints with blend 1 (70 percent PA12), the fracture path goes through the base material, and the fracture starts in the most fragile polymer part—the blend. If the ratio of PA12 in the blend decreases, the weld seam becomes weaker, yet its bonding strength never decreases below that of the pure polymer blend. In general, all the fractures start in the most fragile polymer part. As soon as the fracture reaches the welding seam, the item breaks down. Hence, the joint’s breaking point is not associated with the welding seam.
Conclusion
HDPE and PA12 are typically incompatible for welding. However, either material can successfully be welded to a blend of the two.
Joints of HDPE and its blend can achieve a weld strength close to the tensile strength of pure HDPE (welding factor greater than 0.9). Joints of PA12 and its blends, however, were found to be more delicate when using hot-plate welding.
Although the tensile strength of the involved plastic parts were higher than for pure HDPE, the weld strength could only reach the level of HDPE if the blend had a sufficiently high ratio of PA12 (more than 50 percent). The welding factor remained greater than 0.65. Possible reasons might be the restricted mobility of the modified PA12 chain or the different viscosities of PA12 and the blends. We also identified micro-cracks in the weld seam, which helped diminish the weld strength.
However, the experimental results for diffusion welding show that it is possible to reach high bond strengths for joining PA12 with its blend. In principle, welding factors of 1.0 can be achieved by this method. That means the lower strength achieved when hot-plate welding PA12 with its blend is not a problem of chemical compatibility between the polymers, but rather stems from
the hot plate welding process itself.
Our results indicate the potential of joining any combination of thermoplastics with the aid of appropriate polymer blends, and we intend to test various material combinations in the future.
In addition, our investigations will look into to two-component injection molding for its applicability in large-scale fabrication. The ultimate goal is to join the blend and the base material during injection molding. Thus, the blend may act as an interlayer for subsequent welding processes.
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!



