MEP Aids Medical Device Manufacturers
Many medical device companies have benefitted from the Hollings Manufacturing Extension Partnership

The Massachusetts MEP helped Interscope Inc. develop, manufacture and test a new endoscopic surgery instrument. Photo courtesy Interscope Inc.

Simulation software enabled Paragon Medical to identify and eliminate bottlenecks on its case and tray assembly line. The Indiana MEP showed the company how. Photo courtesy Paragon Medical
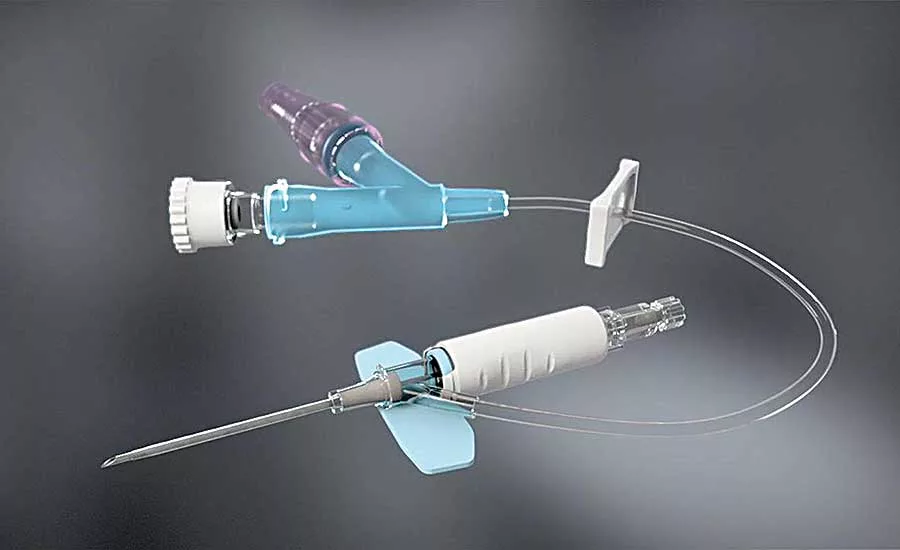
At the Smiths Medical factory in Keene, NH, individual components—tubing, needles, syringes and medications—needed for a procedure are packaged together as one, sterile, sealed kit. Lean training from the New Hampshire MEP helped the company increase output. Photo courtesy Smiths Medical
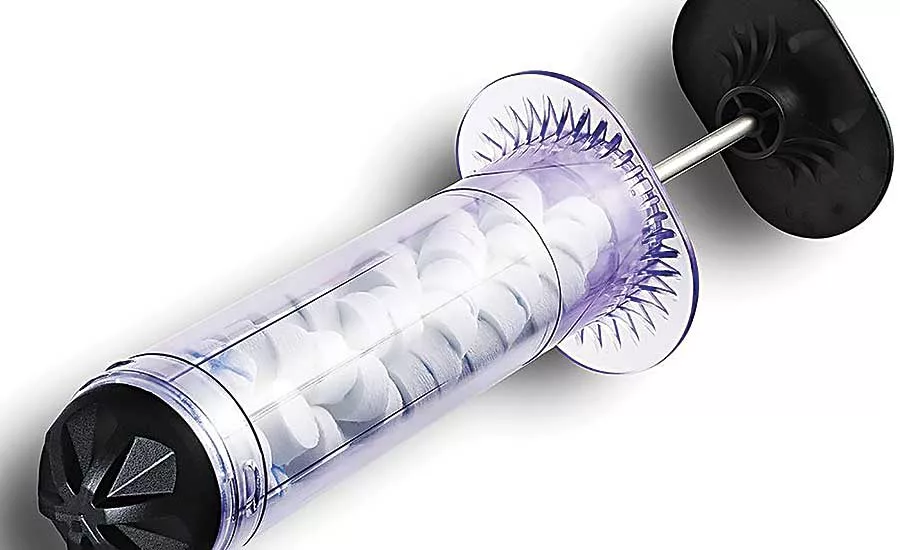
With the assistance of the Oregon MEP, RevMedx automated assembly of its XSTAT product and boosted production capacity by a factor of 10. Photo courtesy RevMedx Inc.

The Maine MEP helped FHC speed development of new, innovative products, like this device for brain surgery. Photo courtesy FHC Inc.
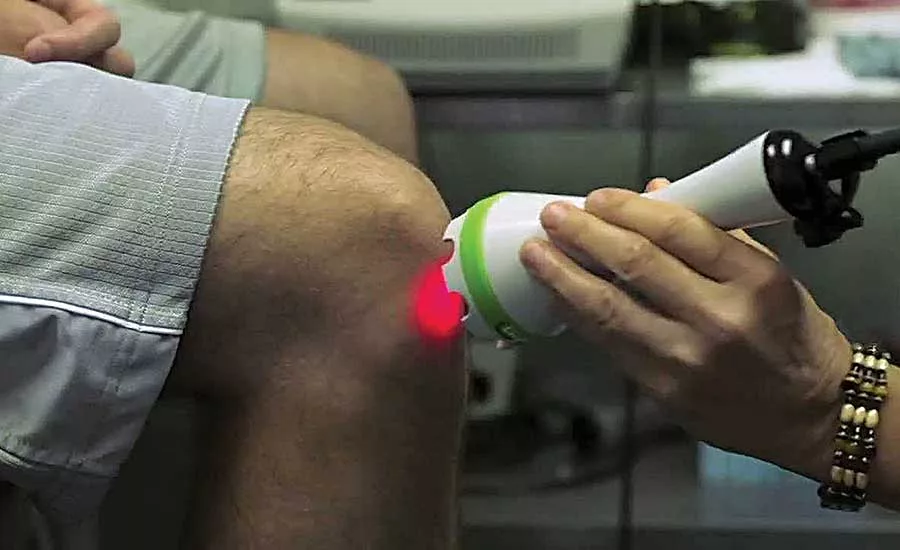
The Delaware MEP helped LiteCure boost export sales of its light therapy devices. Photo courtesy LiteCure LLC
Bringing a medical device to market is a daunting prospect for even the largest manufacturers. There’s nurturing a concept from prototype to finished product; determining how and where to manufacture it; conducting trials for safety and efficacy; and getting FDA approval.
And while the U.S. is home to some of the world’s largest medical device manufacturers, including Johnson & Johnson, Medtronic, Becton-Dickinson and Boston Scientific, more than 80 percent of U.S. medical device companies actually have fewer than 50 employees. Many are start-ups with little or no sales revenue. These companies excel at innovation and product development, but simply lack the manpower to master manufacturing or regulatory issues. A little help can mean the difference between sustained success and going belly up.
Fortunately, small and midsized manufacturers of medical devices—and any other product—have a ready source of help: the federal Hollings Manufacturing Extension Partnership (MEP). The MEP is a public-private partnership dedicated to serving small and midsized manufacturers. With centers in all 50 states and Puerto Rico, MEP assisted 26,313 manufacturers last year, leading to $12.6 billion in sales, $1.7 billion in cost savings, $3.5 billion in new client investments, and the creation or retention of more than 100,000 jobs.
MEP centers and partners have developed a wide range of services and initiatives to help manufacturers identify opportunities to accelerate and strengthen growth and competitiveness in the global marketplace. MEP centers work with manufacturers to develop new products, expand and diversify markets, improve processes, adopt new technology, conserve resources, and enhance value within supply chains. The program also serves as a bridge to other organizations and federal research labs.
As a public-private partnership, the program is delivering a high return on investment to taxpayers. For every dollar of federal investment, the MEP network generates $17.90 in new sales growth for manufacturers and $27 in new client investment. This translates into $2.3 billion in new sales annually. And, for every $1,501 of federal
investment, the network creates or retains one manufacturing job.
Over the years, many medical device manufacturers have benefitted from the MEP. Here are a few of their stories. For more information on the MEP, visit www.nist.gov/mep.
From Concept to Reality
Colorectal cancer is the second most common form of cancer, accounting for more than 150,000 deaths annually. The best way to treat the disease is through endoscopic mucosal resection (EMR), a surgical technique for removing superficial polyps in the gastrointestinal tract before they can grow and become malignant tumors.
Looking for quick answers on assembly and manufacturing topics? Try Ask ASM, our new smart AI search tool. Ask ASM
Interscope Inc., a small medical device manufacturer in Whitinsville, MA, has introduced an innovative endoscopic instrument, called the EndoRotor, to help surgeons completely remove diseased tissue without thermal energy.
Getting to that point wasn’t easy. Back in 2011, all Interscope president and co-founder Jeffery Ryan had were some drawings and an idea for the new surgical device. He didn’t know where to go or what to do to have the product manufactured. Unlike many start-up companies, he was not looking for funding assistance. Instead, he needed help to have proof-of-concept components built to his specifications.
That’s when he called the Massachusetts Manufacturing Extension Partnership (MassMEP).
MassMEP worked with Ryan to develop partnerships with several area businesses and bring his idea from a concept on paper to reality. Algonquin Industries Inc., a machine shop in Bellingham, MA, provided advice on design for manufacturability and produced several sets of prototype parts that enabled Interscope to test and validate the viability of its original concept. Boston Engineering, a product development consulting firm in Waltham, MA, enabled Interscope to expedite R&D. Contract manufacturer Vention Medical of Chicopee, MA, was enlisted to produce the instrument, while consulting firm Icon PLC of Boston helped Interscope meet regulatory requirements for the product. Consulting firm Boston Healthcare provided market research.
The collaborations paid off. Just five years later, in March 2016, Interscope’s EndoRotor was approved for use in Europe, and in May 2017, the device received FDA approval.
“We could not have gotten to commercialization as quickly as we did without MassMEP,” notes Ryan. “Many young companies face business needs that are often not paired well with available resources. The partnership that MassMEP and Boston Engineering presented enabled a dream to take reality in a challenging and dynamic regulatory environment.”
Simulation Improves Flow
Headquartered in Pierceton, IN, Paragon Medical makes surgical instruments (reusable and single-use), cases and trays for those instruments, and implantable components. Founded in 1991, the company has offices and production locations in Indiana, Utah, Switzerland, Poland and China.
Like most manufacturers, Paragon Medical’s Case & Tray Division is faced with the constant need to reduce lead time, lower costs and maintain quality. When ad-hoc projects and localized kaizen events were not achieving the improvement Paragon was looking for, the company contacted the Indiana MEP center, located at Purdue University in West Lafayette, IN.
Staff at Purdue MEP used simulation software from Simio LLC to help Paragon engineers find better processing paths and shop floor layouts. The modeling focused primarily on identifying bottlenecks, rationalizing head count, and reviewing overall product flow. The software also enabled Paragon’s engineers to test proposed improvements so they could identify potential risks prior to full implementation.
The simulation identified three bottleneck areas and recommended combining some process flows for more efficient production. Ultimately, the company saw a 15 percent gain in efficiency.
Paragon even purchased its own copy of Simio’s software for future improvement projects.
“This partnership with Purdue MEP was much more than an academic exercise,” says Sean Miller, director of operations at the Case & Tray Division. “The team engaged in a way that showed their desire to challenge and help Paragon improve our business and operational practices, as is evident in the results we realized.”
Getting Lean in New Hampshire
Smiths Medical manufactures medical devices and procedural kits for use in critical and intensive care applications. At the company’s factory in Keene, NH, individual components—tubing, needles, syringes and medications—needed for a procedure, such as epidural anesthesia, are packaged together as one, sterile, sealed kit.
Ten years ago, the company wanted to bring in a new machine to make hypodermic needles—but there was no room. After assessing the facility, Smiths engineers believed they could reduce the size of the department where pain management kits are assembled. Covering 4,700 square feet, the area contained two large conveyors where employees placed items in kits as they passed by. Engineers wanted to relocate the department and reduce its size by 62 percent.
Engineers also wanted to improve the department’s performance. Batch sizes were getting smaller and the product mix was becoming more diverse. An additional challenge was to increase the number of kits produced per day from 6,800 to 10,700—a 57 percent increase—without adding overtime, increasing costs or decreasing quality.
Smiths contacted the New Hampshire MEP (NHMEP) for help.
The NHMEP believed the plant could benefit from lean manufacturing. A team was created with employees from the first and second shifts of the pain management department. They received basic lean training to become familiar with the methodologies and tools. They also learned how to use value-stream mapping to diagram the current state and determine what improvements were needed to reach their goals.
The team recommended that the large conveyor system be replaced with a cellular layout that used a one-piece, continuous flow. This would enable the department to meet takt times in a substantially smaller space.
The team spent a week with MEP engineers determining the number of cells they would need, how they would be configured, and how many people were needed per cell. In going from one system to the other, the process flow did not change. Various materials are still assembled into trays, and the trays are then wrapped, sealed, and packed. The wrapping and sealing processes do not change from one kit to another, but assembly changes often according to the materials that make up the kits.
The cells were designed according to the takt time of the longest process—wrapping. Four U-shaped cells staffed with six people each would meet their needs. Prior to deployment, the team used tables and benches to test cell arrangements, fine-tune the process, and get used to working in the new cellular configuration. Within a month, the department was operating with a one-piece flow.
“I was skeptical of the manual process after having used the conveyors, but I had an open mind and have been very pleased with the results,” says Jeff Rondeau, senior business unit supervisor at Smiths Medical. “The employees have a better attitude. They work in smaller groups, and both the quality and output are better. We are much more flexible now!”
Standard work procedures were created for every process in the department, which are helpful in training new employees.
A staging area was constructed between the drug storage area and the pain management department. There, orders and materials for the kits are pulled onto wheeled racks, which are moved to the cells when visual kanbans indicate the need. Three computer workstations show bills of material with photos for each order. Materials are pulled according to photos, and a second copy travels with the work order to the workcells for quality control.
The staging area allows materials and drugs to be filtered into the line simultaneously and more efficiently. Setup reduction resulted in a typical kitting time of 30 minutes, down from the original 45 minutes.
Workers did not require much training to work in the cellular layout, though they did need to get comfortable having just 18 seconds to handle seven components under the new system. Inspection is done at the source, so quality rests on the workers.
Everyone works together as a team. Line balance encourages teammates to help each other. Line leaders at the front of each cell set the pace and can hop over to assist the wrappers at the end of the process if they get bogged down. Workers known as “water spiders” assist with supply replenishment when the cell is fully staffed.
Thanks to the cellular arrangement, the department is now able to complete four kits every 18 seconds. Using the same number of employees and changing from two conveyor lines to four workcells, the department:
- Reduced floor space by 62 percent.
- Increased output per shift by 82 percent, from 2,720 to 4,950 trays.
- Increased units output per hour of labor from 7.2 to 14.4.
- Increased the average number of changeovers from 15 to 40.
Smiths Medical has since been applying lean principles in other departments at the factory. In addition, more than 20 manufacturing and support staff from the facility have gone through a lean manufacturing certificate program at a local community college.
Automating in Oregon
“Bleeding out” from wounds on the battlefield is the leading cause of death for soldiers. Medical device manufacturer RevMedx Inc. of Wilsonville, OR, was formed in 2010 by a group of combat veterans, scientists and engineers to solve that problem. The company’s flagship XSTAT product can be rapidly deployed where it may be impossible to put on a tourniquet or apply manual compression externally, providing fast-acting hemorrhage control to stabilize a wounded patient for transport.
The XSTAT was granted FDA approval in April 2014. Today, the company offers a suite of products that work in conjunction with the XSTAT, and it continues to conduct research on new medical products.
Early acceptance of the XSTAT product by the military, emergency medical services, and law enforcement agencies required RevMedx to expand its production capabilities. In addition, the company needed to reduce the cost of producing the device to make it more affordable.
RevMedx contacted the Oregon MEP (OMEP) for help.
To scale up manufacturing of the XSTAT, OMEP worked with systems integrator Automation Solutions of Beaverton, OR, and MilTech, a Defense Department agency that helps accelerate private sector development of new technology for the military.
RevMedx personnel collaborated with OMEP, MilTech, and Automation Solutions to develop automated equipment that would meet the company’s needs. After identifying new, high-volume production processes requiring unique specialized equipment, the team performed proof-of-concept testing and developed custom machine specifications.
The XSTAT product works by injecting a large number of small, rapidly expanding minisponges into a wound cavity using a syringe-like applicator. The compressed minisponges expand upon contacting blood and swell to fill the wound cavity, providing internal pressure to slow down or stop internal hemorrhaging within 20 seconds.
The new machines automate production of the minisponges using a combination of robots, vision systems and other advanced processing techniques.
As a result of its collaboration with OMEP, RevMedx has added jobs, lowered costs, improved yields, and boosted production capacity by a factor of 10.
Filling the Product Pipeline in Maine
FHC Inc. of Bowdoin, ME, manufactures microelectrodes and special-purpose instruments for neurosurgeons. A key factor in the company’s success has been its entrepreneurial zeal. Since its founding in 1970, FHC has designed and manufactured more than 7,000 unique metal microelectrodes and a broad range of state-of-the-art clinical and research instrumentation.
Recognizing that innovation is critical for future growth, the company historically has had multiple potential new products in the pipeline. But, like many small companies with limited human and financial resources, many of these projects remained unfinished. To address this problem, FHC engaged the Maine MEP to provide innovation engineering coaching and lean manufacturing consulting.
The Maine MEP recommended an “innovation engineering” program. Typically one to three months in duration, the program consists of three steps: education and immersion by an MEP project manager in a company’s challenge, a one-day innovation “jump start” session, and project acceleration coaching.
FHC used the jump start to successfully address a challenge encountered by the company’s European customers, who prefer to use a shorter lead on FHC’s microTargeting and Star Drive neurosurgical devices. Having seen how the techniques of innovation engineering could be used to modify a product, prototype a design, and commercialize it, FHC decided to embrace the process on a wider scale.
Speed and efficiency are two advantages of innovation engineering. “It provides a structure for out-of-the-box thinking, and yet balances that creativity by providing guidelines that require focused energy and timelines, while curtailing waste,” explains John Karp, technology accelerator program manager with Maine MEP and entrepreneur in residence at the Maine Technology Institute in Brunswick, ME.
The initiatives with Maine MEP have directly impacted FHC’s bottom line. Just two years after implementing the innovation engineering program, FHC was able to retain some $500,000 in sales, 25 percent of which were due to new products filling in for some going obsolete. FHC also credits the program for $250,000 in cost savings and $240,000 in new investment, and it has seen a marked increase in employee participation in new idea generation.
“Innovation is the key to growth in today’s global market. But it’s important to know not just how to innovate, but how to bring those new products to market with minimal risk,” says Karp. “Innovation engineering gives firms the strategic tools to assess the market, gauge demand, prototype rapidly, and cost-efficiently commercialize new ideas. It’s an approach to product development that every Maine manufacturer should embrace.”
Improving Exports in Delaware
LiteCure LLC of Newark, DE, makes devices that use light to alleviate pain or inflammation, boost the immune system, and promote wound healing and tissue regeneration. The devices are used to treat both people and animals.
A few years ago, when LiteCure was looking boost sales in international markets, the company enrolled in an ExporTech program delivered by the Delaware MEP (DEMEP). ExporTech is a national program created by the U.S. Department of Commerce and administered by MEP centers to help companies strategically plan for developing business in internal markets.
Throughout the year, DEMEP partners with numerous export resources to co-host ExportTech events in the state. These resources include the U.S. Export Assistance Center—Philadelphia, Global Delaware, World Trade Center—Delaware, the Small Business Administration, FedEx and Fulton Bank. These strategic planning sessions help companies find the right markets for their products and develop roadmaps for entering those markets with optimized results.
After participating in ExporTech, LiteCure saw $130,000 in sales growth in Denmark and $275,000 in sales growth in Korea. The company also developed a strategy for market penetration in Germany.
“Learning from experts showed us what we did not know. We had been fairly successful in export sales prior to taking the class. Getting practical advice on contract structure, financing options and marketing strategies helped us get more effective and professional. We would recommend this course to any company looking to accelerate sales overseas,” says Andy Wood, vice president of international sales for LiteCure.
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!




