Automated Assembly of Medical Devices
Sharps, test cards, sensors and miniature gas canisters are among the many medical devices produced on automated assembly systems

This automated assembly system fills and welds tiny stainless steel gas cylinders. Photo courtesy Mikron Corp. Denver

Vision-guided robots perform a variety of tasks in this assembly system. Photo courtesy RND Automation

This lean cell assembles a noninvasive surgical device every 15 seconds. Photo courtesy Invotec Engineering Inc.

An automated transfer system places contact lenses into blister packs at rate of more than 12,000 pieces per hour. Photo courtesy Edgewater Automation

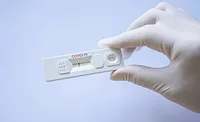
This flexible assembly machine produces flu cards. It can accommodate 10 variants. Photo courtesy Lanco Integrated

This cam-driven assembly system produces approximately 1,000 needle assemblies per minute. Photo courtesy The Arthur G. Russell Co.
Disposable devices are essential components of every medical, dental and veterinary practice. Using disposables lowers costs, increases efficiency, and reduces the spread of infection. According to market research firm The Freedonia Group, sales of medical disposables in just the U.S. are expected to top $49.3 billion this year.
Catheters, syringes, inhalers, testing supplies and other disposables must be produced by the millions annually. Meeting that kind volume can only be accomplished with automation. As the examples below illustrate, systems integrators have designed and built some innovative machines for medical device assembly.
System Assembles Tiny Gas Cylinders
Based in Golden, CO, PICOCYL designs and manufactures very small, compressed-gas cylinders for powered drug-delivery systems, self-injection devices, automated implant insertion, and the delivery of medical gases. The company’s proprietary, serialized “pico-cylinders” feature accurate and consistent gas fill levels, low puncture forces, precision sealing surfaces, and stainless steel construction.
The company’s products are manufactured, filled and sealed with a state-of-the-art automated assembly system in an ISO Class 8 clean room in the U.S. The assembly system was designed and built by Mikron Corp. Denver.
A pico-cylinder is approximately 8 millimeters in diameter and 27 millimeters long. The cylinder consists of two parts—a canister body and a cap—and is filled with carbon dioxide.
The assembly system is based on Mikron’s G05 platform and consists of two linear indexing cells that synchronously transfer assemblies between process stations. The cylinders are fixtured on pallets measuring 120 millimeters square.
Bulk hoppers meter parts to vibratory bowls that discharge oriented parts to inline tracks and escapements.
Looking for quick answers on assembly and manufacturing topics? Try Ask ASM, our new smart AI search tool. Ask ASM
Resistance welding is used to secure the cap to a canister body after filling. To ensure quality, the weld current is monitored during assembly. After welding, a linear variable differential transformer (LVDT) verifies the overall height of the assembly, and a precision scale weighs each cylinder.
A laser marks finished components with the fill weight, lot number and the end-customer’s logo.
The system produces 18 parts per minute.
Mikron worked closely with PICOCYL to define parameters for optimal process reliability and to maximize system throughput. Maintaining tight tolerance for canister fill weight with a repeatable process was a challenge, says Vincent Mettraux, project manager at Mikron. Process controls were developed for monitoring the temperature of the bulk CO2 fill tank, as well as regulating gas pressure at the fill stations based on feedback about the weight of finished and filled cylinders.
The system accommodates a single product variant, but it can be configured to assemble and fill multiple components in parallel or to fill the cylinders with other gases.
Over the years, Mikron has developed and built multiple systems with different requirements for PICOCYL. “Developing [an] innovative medical delivery system using a gas-powered cylinder for injection force required new manufacturing processes and a collaborative partnership to automate them,” says Bob Barron, CEO of PICOCYL. “As a startup company, this was no easy task. Mikron believed in our vision and stood by our side to enable our success. We are now on our fourth Mikron automation system.”
For more information, call Mikron at 303-364-5211 or visit www.mikron.com/automation.
System Refurbishes, Packages Oxygen Sensors
A pulse oximeter is a small, noninvasive, photoelectric device that attaches to a fingertip with pressure-sensitive adhesive. The device measures the amount of oxygen in the blood by detecting differences in how light is absorbed by oxygenated or deoxygenated blood. It can rapidly detect even small changes in how efficiently oxygen is being carried to the extremities furthest from the heart.
Hospitals use hundreds of these sensors daily. The devices aren’t cheap, and since they’re noninvasive, they can be cleaned, checked, equipped with fresh adhesive, and reused multiple times.
Recently, systems integrator RND Automation & Engineering LLC designed and built a machine to automate the process of refurbishing and repackaging pulse oximeters. The system is a servo-powered rotary indexing dial machine with 12 stations.
The oximeter consists of an adhesive-backed woven fabric patch, medical tape backing, LED sensors, a release liner, wires to connect to the sensors, and a connector plug with an embedded memory chip.
To start the process, the fabric patch is measured, cut, and placed onto a fixture on the dial. An actuator mechanism then removes the paper backing from the patch to expose the adhesive. Next, an operator loads the sensor and wires into a fixture.
At the next station, a custom roller mechanism wraps the fabric patch around the cord. At the next two stations, a vision-guided SCARA robot places tape on the sensor, and another vision-guided robot conducts a functional test of the device. The machine uses a SmartSAT analyzer from Clinical Dynamics Corp. to test the operation of the LED sensors. The machine also performs a continuity check on the sensor and resets the device’s memory chip to factory standards.
With that done, a release liner is added to the assembly, and a laser marker places a 2D matrix code on the connector if one does not already exist. Most of the adhesive-backed parts are fed by label applicators and presenters with customer modifications.
At the end of the line, a six-axis robot places the final product into a winding station, which neatly coils the oximeter’s wires. Then, an X-Y-Z gantry system loads the entire sensor assembly in a Tyvek pouch approximately 6 inches long and 4 inches wide. Finally, a Kanga pouching machine—a standalone product developed by RND Automation for medical device packaging—seals the pouch.
The machine uses multiple vision system cameras to locate and align the two sensors on the product, as well as to verify placement of the medical tape. The system also uses multiple barcode scanners to check the number of times the sensor has been refurbished and to track UDI requirements.
The system produces one part in less than 6 seconds. Three different sensors can be run on the machine, and changeover is entirely automatic. “The product was not designed for assembly,” notes Sean Dotson, PE, president and CEO of RND Automation. “The sensors vary vastly in size, shape and cord memory.”
The stickiness of the parts was a challenge. “The product is both sticky and has varying shapes due to the memory in the cord,” Dotson explains. “We had to develop a way to allow sticky parts to be held with vacuum, yet release from the fixtures when complete. A special metal coating was used to prevent the adhesive from sticking to the metal fixtures. Cord management was also important to prevent snags or parts being pulled out of the nests.”
RND built two systems in parallel. The company produced all aspects of the machine, from loading to final pouching.
To see a video of the system, click https://tinyurl.com/y7zx2yfo.
For more information, call RND Automation at 941-870-5400 or visit www.rndautomation.com.
Cam-Driven System Assembles Sharps
Cam-driven indexing systems have numerous advantages. They are reliable, durable and easy to run. Cam-driven systems provide controlled accelerations and decelerations; accurate and repeatable positioning; high load and speed capability; and smooth motion.
Those advantages make cam-driven systems ideal for assembling small products—like medical disposables—at high volumes. Systems integrator Arthur G. Russell Co. (AGR) recently built just such a system to produce a needle-based medical device. The device is approximately 0.35 inch in diameter and 2.1 inches long. It has five parts, including a stainless steel needle and a label.
The system consists of a cam-driven synchronous line integrated with a continuous-motion dial platform. Multilane vibratory bowl feeders provide a steady stream of parts to the line. The feeders are governed by AGR’s modular VibroBlock system, which allows engineers to fully adjust the vertical and horizontal components of the vibratory motion.
“Our engineered feeders can deliver parts at much higher rates,” says David Aquilino, customer service and marketing manager at AGR. “Another important feature of the VibroBlock system is the controller, which uses state-of-the-art electronics to vary the duration of the energy pulses applied to the driving unit. This permits higher efficiency and much closer control of amplitude than is possible with variable-voltage controllers.”
An adhesive is used to bond the needle to the plastic hub, and high-end vision systems and photoelectric sensors are used for inspection and error-proofing.
The system assembles approximately 1,000 devices per minute, and the system can accommodate variations in the gauge and length of the needle.
For more information, call AGR at 860-583-4109 or visit www.arthurgrussell.com.
Lean Cell Blends Manual, Automated Assembly
Assembly lines for medical devices don’t have to be entirely automated. In some cases, a mix of manual and automated stations is more efficient and cost-effective.
Recently, systems integrator Invotec Engineering Inc. designed a lean cell to produce a noninvasive surgical device. The challenge of this line was to balance the simplicity of manual stations with the complexity of fully automated stations—leveraging the benefits of both to create reliable, repeatable results. The assembly line consists of approximately 15 stations. Operators perform one or two tasks at each station and transfer work-in-process manually.
The final product is approximately 0.25 inch in diameter and comes in three lengths: 40, 45 and 50 centimeters. The device has 25 parts made from stainless steel and various polymers.
Most of the subcomponents are manually assembled with presses and custom fixtures. To prevent errors, vision systems are used throughout the line to confirm part presence, orientation and location. Vision systems and LVDTs are also used to measure the assemblies. Parts that are too difficult to handle are fed by five feeder bowls. The system also includes six laser welding systems and a heat-staking machine. The laser welders are also equipped with vision systems for accuracy, inspection and weld quality analysis. Leak testing, strength testing and pull testing are all conducted on the line.
The system started out with a feasibility study and grew into a full production line. Invotec ended up developing the equipment in parallel with the customer. “The customer was finalizing the product design while we manufactured the equipment,” says Isabel Caldwell, applications engineer. “To accommodate design changes, our customer was given full access to the equipment throughout the process. This allowed both teams to better understand how the system will function in production and what challenges might arise in that setting.”
For more information, call Invotec at 937-886-3232 or visit www.invotec.com.
High-Speed System Packages Contact Lenses
Some 45 million people in the U.S. wear contact lenses. Producing them requires some very high-speed automation, indeed.
Recently, Edgewater Automation SC designed and built a high-speed transfer machine capable of picking contact lenses out of a saline solution and placing them into blister packs at rate of more than 12,000 pieces per hour. The machine fills each blister pack with saline solution, picks and places the lenses, and transfers the packs to pucks without spilling a drop of the solution.
Approximately 20 feet long and 20 feet wide, the conveyor-based system uses pallets and pucks to transport the contacts from station to station.
The blister pack is 1.5 inches long and 0.5 inch wide. The packs are fed using a vibratory bowl. The lenses are fed using a tray system. The pick-and-place system uses a vacuum head to pick up the lenses.
A vision system with ultraviolet lighting ensures that 99 percent of the lenses are picked and placed correctly.
“To avoid spilling the saline solution, we were able to fine tune the Allen Bradley Kinetix servos to achieve the fastest possible acceleration and deceleration,” says Jamen Blake, simulation engineer at Edgewater. “Once you’re at max speed the solution won’t spill, so it’s all about how you accelerate and decelerate.”
Preventing corrosion from the saline solution was a design factor. “We had to make this machine IP68-rated, which means it can be washed down,” says Blake. “All the tooling was made out of 316 stainless steel, aluminum and Sustarin medical-grade acetal plastic. This ensured no part of the machine would be susceptible to corrosion and bacteria.”
For more information, call Edgewater Automation at 864-381-6122 or visit www.edgewaterautomation.com.
System Assembles Flu Cards
The flu is a serious illness. During the 2014-15 flu season—one of the worst in history—some 34 million Americans contracted the disease, 710,000 were hospitalized and 56,000 died.
Determining exactly which strain of the flu virus is causing someone’s illness is vital not just for treatment, but for formulating an effective vaccine for the following year’s flu season. That’s what flu cards are for.
Machine builder Lanco Integrated designed and built a fully automated assembly system to produce flu cards. Based on the company’s HFL 2002 pallet-transfer system, the line produces 6,000 cards per hour. The flexible system can run 10 part variants. Changeover is achieved via the machine’s HMI.
Although the card consists of seven components, only one operator is needed to keep the machine supplied with parts.
Components are automatically fed to the system through card and reel feeders. Because of the inherent variability of the parts, and because components must be placed precisely, “on-the-fly” vision feedback is necessary to ensure accurate placement. Throughput is supported by two-up reel feeding, allowing for easy changeover of one reel while the other continues feeding the line.
The system can produce four flu cards per pallet.
For more information, call Lanco at 207-773-2060 or visit www.lanco.net.
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!