Surface Preparation and Medical Silicone Adhesives
Cleaning and priming substrates prior to bonding can significantly improve adhesion


Medical device manufacturers often use silicone adhesives to assemble products such as catheters, pacemakers, cochlear implants, aesthetic implants and gastric balloons.
In addition to evaluating different adhesive formulations, device manufacturers need to critically assess several other factors in the adhesion process, including the substrates to be bonded, their surface energy and even the overall device manufacturing process. Without addressing these factors early in the design process, device makers risk costly delays and potential manufacturing issues.
Medical devices may consist of multiple parts that must be permanently joined and sealed. In most cases where silicone adhesives are used, the adhesive is the sole element that bonds two surfaces together. In other cases, adhesives may be used in conjunction with a mechanical element to join two surfaces, such as a slot and groove, a pin and hole, or other physical joining component.
In general, most medical devices, whether implanted, inserted or used externally, are made of one or more of the following materials:
- Silicone.
- Metals, including aluminum, stainless steel and titanium.
- Plastics, including polytetrafluorethylene (PTFE), polycarbonate, polyurethane and polyimide.
An increasing number of medical devices, such as oxygenators and blood reservoirs, are made with polycarbonate due to its ease of molding and light weight. Another plastic, polyamide (often known as nylon), can usually be found in common products such as medical tubing and catheters. Titanium and medical-grade silicone rubber are often used for many implanted medical devices, such as pacemakers, cochlear implants, hydrocephalus shunts and implantable infusion pumps. In some cases, two pieces of the same substrate—plastic, silicone or metal—are sealed with silicone adhesive. In other cases, two different types of material—such as silicone to metal or silicone to plastic—need to be joined and sealed.
Depending on the substrates being adhered, simply applying the silicone adhesive may not provide the desired level of bonding. Device manufacturers might also discover that the substrate materials they selected, such as PTFE, may be very difficult to bond using silicone adhesives without first doing some type of surface treatment.
Adhesion must be absolutely secure to preserve the long-term functionality of the device, especially if it is implanted in the body. Many medical devices are inherently small, and their joints are also quite small, requiring careful manual work. If it becomes difficult and time-consuming to achieve a proper and complete seal, throughput can suffer. Additional inspection steps or rework of seals may need to be done if substrates fail to fully accept the adhesive application.
With the numerous substrates available and multiple adhesion tests to potentially choose from, there may be challenges with expecting a supplier to have data specific to your application. A custom formulation may also be required, so it’s important to work with a capable silicone adhesives manufacturer early in the process to discuss critical factors. To achieve reliable adhesion, three main factors should be addressed, preferably during the device design stage:
- The “wettability” or surface energy of the different substrate materials being adhered.
- Surface cleaning to avoid inhibiting adhesion.
- The use of appropriate primers to better prepare surfaces to fully accept silicone adhesives.
Understanding Surface Energy
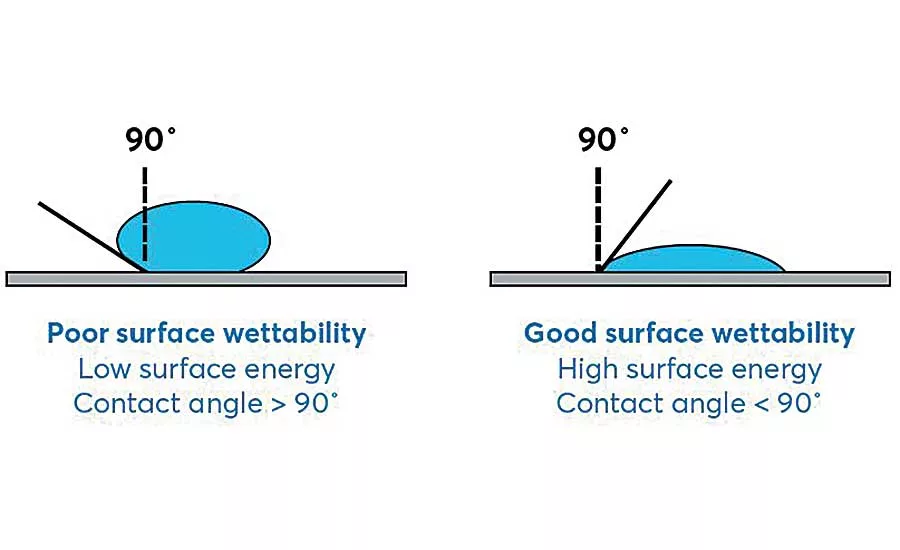
The receptiveness of a substrate to an adhesive is generally related to its surface energy, or “wettability,” which refers broadly to substrate acceptance of a coating. Low-surface-energy materials, like polyethylene, do not allow a liquid adhesive to easily “wet out,” or spread outward, across their surfaces.
Looking for quick answers on assembly and manufacturing topics? Try Ask ASM, our new smart AI search tool. Ask ASM
The contact angle of a drop of water on the substrate surface helps quantify the wettability of the surface. In general, if the contact angle is below 90 degrees, the material has high surface energy and provides good surface wettability. If the contact angle is greater than 90 degrees, the material has low surface energy, with a greater likelihood of adhesion failure due to blank spots on the substrate surface where no adhesive is present.
Plastics, such as polyethylene, have very low surface energies, which can make it challenging to use silicone adhesives without proper surface preparation. Better wetting also enables greater interaction with the substrate. The silicone can more easily fill in the microscopic valleys on the surface to provide better adhesion.
Cleaning, activating or priming a substrate’s surface can increase wettability and maximize available bond sites to improve adhesion. When developing a surface preparation procedure, there are several options to consider.
Topping the list is substrate cleaning. Proper cleaning methods remove contaminants, such as finger oils, dust particles, mold release agents and machine oils on metal parts. Surface cleaning can be done by mechanically or manually wiping the surface with a solvent, such as isopropyl alcohol or a heptane, using swabs or lint-free cloths.
Surface treatments are another option. To help increase the substrate’s surface energy for better adhesion, several methods can be considered. Plasma treatment bombards the substrate surface with ions of a gas, such as argon. Corona discharge is popular, because it can be done in-line during automated production. During corona treatment, electrons accelerated onto the surface of the plastic create oxygenation, which generates new carbonyl groups with a higher surface energy. This increases reactivity, allowing the adhesive to spread over a larger area rather than bead up.
A third option is a primer. These materials act as coupling agents, increasing and strengthening the covalent bonds between the adhesive and substrate. They have also been shown to greatly increase the silicone adhesive’s ability to wet out the substrates. Note that there is no “one-type-fits-all” formulation for primers. The substrate, the adhesive, the manufacturing process and even certain regulatory requirements all play a role in determining the best primer for a given application.
Testing Surface Preparations
Each substrate being joined using silicone adhesives has a unique surface energy. To ensure that complete and effective adhesion is achieved during production, engineers should accurately characterize and quantify the surface energy of the substrates. With this data, they can make informed decisions about surface cleaning, surface preparation and the right surface primer to use—decisions that must be made before device designs are completed and production and assembly processes are finalized.
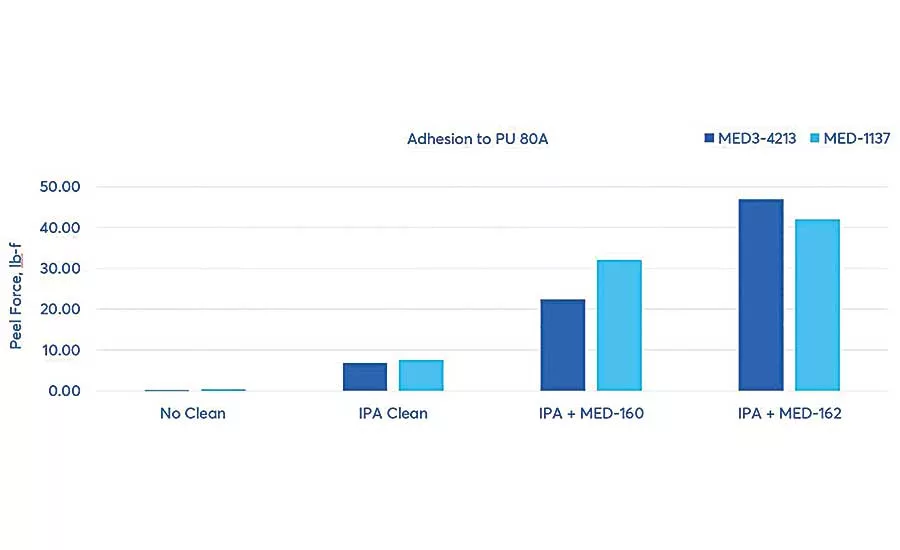
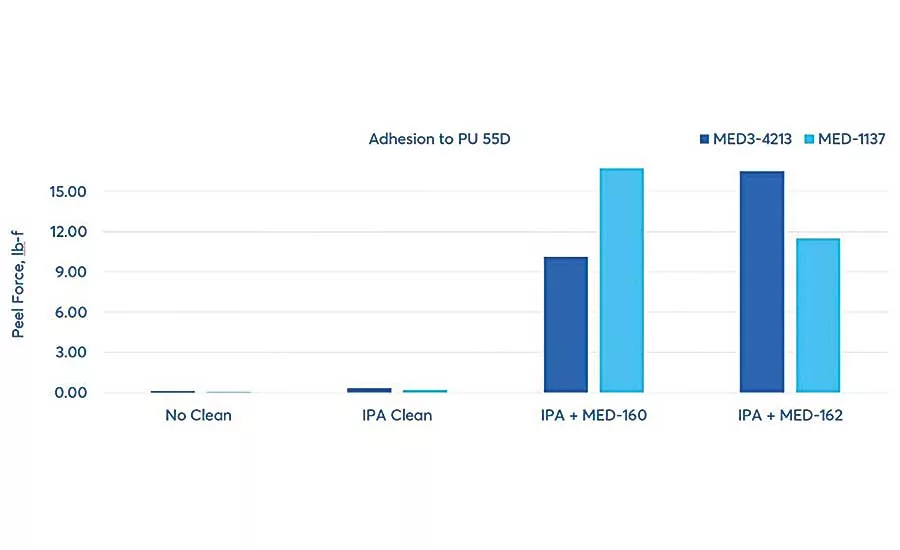
To evaluate how adhesion can be improved through these surface preparation steps, NuSil silicone adhesive experts conducted tests to demonstrate the effectiveness of substrate cleaning and adhesive primers. The test involved two substrates: Pellethane 2363 80A, an 80 Shore A durometer thermoplastic polyurethane elastomer, and Pellethane 2363 55D, a 55 Shore D durometer thermoplastic polyurethane elastomer. The adhesives were MED-1137, a one-part, tin-catalyzed, room temperature vulcanizing (RTV) silicone adhesive, and MED3-4213, a two-part, platinum-catalyzed, fast-curing silicone adhesive. The primers were MED-160, a general-purpose primer used with moisture- and platinum-cure silicones, and MED-162, a primer specifically for use with polyurethane, polycarbonate and polyetheretherketone (PEEK) and platinum-cure adhesives.
Sample preparation—cleaning, priming and adhesive application—was conducted at room temperature (20 to 26 C) and 40 to 60 percent relative humidity. All samples were placed in a controlled environment chamber set at 24 to 26 C and a relative humidity of 40 to 60 percent. Curing times were set according to each adhesive’s design specification.
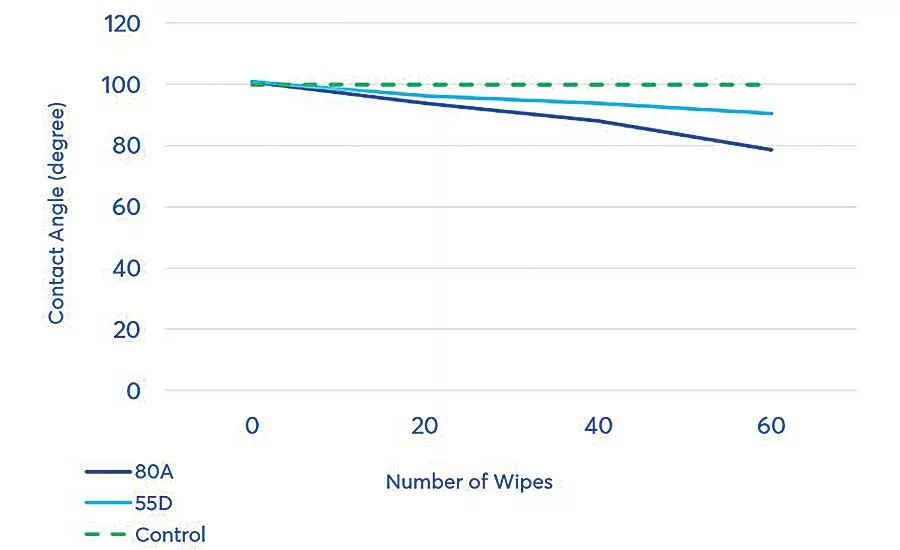
The first surface preparation factor studied was the effect of surface cleaning on surface wettability—specifically whether, and to what extent, multiple cleaning passes affect the contact angle on the two polyurethane substrates.
The surfaces of both substrates were wiped using isopropyl alcohol with a mechanical wiping device to eliminate inconsistencies in manual wiping, such as pressure against the substrate and area to be cleaned. The contact angle was measured after 20, 40 and 60 wipes and compared to the contact angle without cleaning. Results showed that cleaning the surface significantly improves the contact angle.
Next, the primers were applied to the newly cleaned polyurethane substrates. For each substrate, the peel force required to separate the polyurethane panel from the silicone was tested, assessing the effect of surface cleaning and the use of adhesive primers.
The first test assessed adhesion and the effect of surface treatments on Pellethane 2363 80A. The polyurethane panel and a sheet of silicone were adhered using MED3-4213 and MED-1137 silicone adhesives with and without the use of the two primers. Peel force tests were conducted using an Instron 3345 tensile tester set at a pull rate of 20 inches per minute, and the peak force was reported. Significant improvements were shown with surface cleaning alone, and even more so when primers were used. MED-162 primer provided the most effective improvement in adhesion for both silicone adhesives tested.
The tests were repeated on the Pellethane 2363 55D, using the same surface cleaning, silicone adhesives and surface primers. For this substrate, although only slight improvements in peel force were shown with surface cleaning alone, significant improvements were shown through the use of both primers. The combination of MED-160 primer and MED-1137 adhesive outperformed all other combinations.
Final Factors for Best Results
Silicone adhesives are a trusted and widely used material for many types of medical devices. These results demonstrate that for the specific polyurethanes tested, both surface cleaning and the use of primers significantly improve adhesion. Our tests show that medical device manufacturers using silicone adhesives can benefit from assessing the surface energy or wettability of substrates they are joining with silicone adhesives.
Along with choosing the best silicone adhesive for a given device, manufacturers should take steps to ensure that they understand the surface energy of the substrate so that it can be effectively prepared and suitably wetted to ensure maximum adhesive dispersal and reliable adhesive performance. Because of the variety of adhesives, substrates and manufacturing processes, there are advantages to working with an adhesives supplier that can characterize and perform tests to quantify adhesion requirements specific to the application. Adhesive suppliers can also supply insight on the appropriate silicone adhesive and primer to use for the specific application. In some cases, due to unique requirements, a special formulation may be required. It is beneficial if the supplier is equipped to develop and manufacture custom adhesives.
Finally, with silicone adhesives and primers used for medical devices, it’s especially important not to overlook a supplier’s manufacturing and purity qualifications, as well as support during the regulatory process. An adhesive supplier that can provide master files to the U.S. Food and Drug Administration and international authorities to document product details regarding ingredients, manufacturing, processing, packaging and storage can save a device maker valuable time when taking the final product to market.
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!