Medical Device Manufacturers Require Tighter Leak Test Requirements
Tighter leak rate requirements are driving demand for smarter equipment in medical device manufacturing.
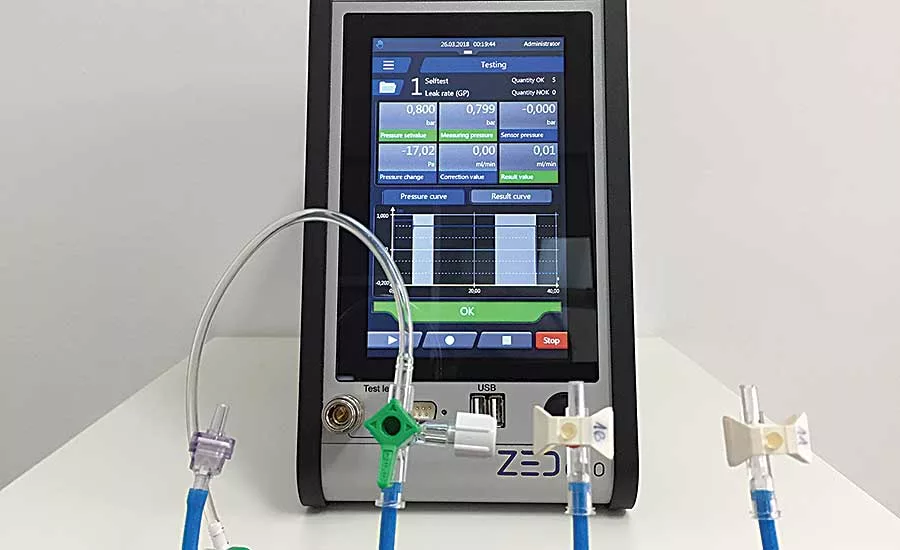
State-of-the-art leak testing equipment features data traceability functions. Photo courtesy Zeltwanger Leak Testing & Automation LP
Medical devices can be difficult to check for leaks, because they often incorporate flexible components. Photo courtesy Vesta Inc.

This compact device monitors flow and pressure of medical devices with fast response time. Photo courtesy InterTech Development Co.

This helium leak detector is flexible and can handle multiple test methods, such as sniffing, chamber testing and helium bombing. Photo courtesy LACO Technologies Inc.
Catheters, pacemakers, ventilators and dialysis filters are vastly different products. But, they all have one thing in common: they must be checked for leaks, whether into, or out of, an assembly.
Medical devices often form a barrier between fluids or gases that could create dangerous conditions for patients if they were allowed to mix. Some products are the route for delivery or extraction of fluids.
If a leak along the route exists, then the fluids could be delivered to the wrong part of a patient’s body or bodily fluids could gather inside the body. Complete or partial blockage of medical devices could have fatal consequences, as could the failure of a device to deliver the correct dosage.
Because many medical devices have fluid management functions that ensure safety and reliability, engineers demand strict parameters for leak testing equipment.
“A leaking medical device can cause dire consequences, unlike a product like a waterproof speaker, where a leak just creates a minor inconvenience,” says Jim Burch, sales engineer at ATEQ Corp. “The compressed air supply used for leak testing needs to be of a higher quality than air used to test products such as automotive components or irrigation valves.”
“Medical devices have the absolute highest demand on quality, functionality and cleanliness,” adds Juergen Goehner, president of Zeltwanger Leak Testing & Automation LP. “With medical devices, the product is commonly used inside or in direct contact to human bodies.
“All devices need to work 100 percent of the time, error free, day after day,” explains Goenher. “There is no grey zone; only black or white.
Looking for quick answers on assembly and manufacturing topics? Try Ask ASM, our new smart AI search tool. Ask ASM
“Every part needs to be fully tested and traced,” Goehner points out. “And, the wide range of devices in use—each one with different characteristics—requires a testing platform that can accommodate the varying parameters and needs of each device, yet still provide a common user interface.”
“The leak testing market for medical devices is very diverse,” says Chuck Davidson, business development manager at ATC by Pfeiffer Vacuum. “[It ranges] from drainage systems, catheters and neuro valves all the way up to medical implants, such as pacemakers.
“There are different requirements and trends connected with each of those product categories," notes Davidson. "Generally speaking, the need for 100 percent testing, and therefore, higher cycle times, is connected with increasing sensitivity demands. In the field of helium leak detection, we also see a trend to “dry” (oil free) systems to avoid any possible contamination.”
“Because of new materials, there are many soft, flexible products now being made,” adds Dan Hoffman, production leak detection sales and service manager for the Americas at Inficon Inc. “More medical devices today contain electronics and sensors that must be isolated. Tightness is more important than ever. That’s driving the need to find increasingly smaller leaks and imperfections.”
To address those issues, medical device manufacturers typically specify nondestructive test methods that can evaluate the integrity of a product for significant leaks within short cycle times. They are looking for leak testing equipment that’s smart, easy to clean, easy to operate and can handle multiple data collection scenarios.
Testing Variety
Leak testing verifies the integrity of a manufactured product. Tests range from pressure testing and pressure decay to destructive burst testing and bubble break point testing.
Testing equipment can be used to detect a wide variety of problems and flaws, such as gross leaks, fine leaks, blockage, cracks in seals, crimped lines, rolled seals, kinked hoses, and defective valves and pressure regulators.
There are two basic methods used to test products for leaks. In one method, the product is pressurized, isolated from the pressure source, and observed to detect escaping gas. Bubble, sniff, pressure decay, differential pressure, mass flow and mass spectrometry testing fall into this category.
In the other method, the product is evacuated and then monitored to see if gas flows into it. Pressure rise and mass spectrometry are in this category.
Many different types of fully programmable high-pressure leak testing devices are available, such as hydraulic pressure testers and gas leak pressure testers.
Hydraulic pressure testers use distilled water as the pressure medium. These devices are used for burst testing, fatigue testing and compliance measurement of medical tubing, plastic fittings, polymer balloons and other components.
Gas pressure leak testers operate with pure dry nitrogen. They are typically used for final product inspection, because the nitrogen gas does not contaminate the product.
Pressure decay or mass flow using air is the preferred method for leak testing in the medical device industry. With pressure decay leak testing, a device is pressurized, isolated from the pressure source, and monitored to see if the pressure decreases.
Because air moves from a high-pressure area to a low-pressure area, a leak path can be detected in the chamber. Any reduction in pressure over time is displayed as a pressure change rate. Or, the pressure reduction can be converted by calculations to get the volumetric leakage rate.
In differential pressure leak tests, a reference volume is pressurized along with the test part. A transducer reads any pressure differential that develops over time between the leaking part and the nonleaking reference.
Differential pressure testing is well-suited to applications requiring relatively high test pressures. And, it typically provides higher sensitivity, more repeatability and faster test times than pressure decay testing.
Traditional pressure decay leak testing measures the pressure at two points in time. However, these simple measurement systems can be prone to error.
The problem with this type of analysis is that it relies on only two data points. Reading electrical transducers always has some degree of uncertainty, due to electrical noise and other inaccuracies.
Signature analysis is a more accurate test method. Signature analysis tools capture leak test waveforms in real time and then apply algorithms to automatically analyze the entire leak curve instead of just measuring two points in time. Using all the data points in the curve leads to increased accuracy, and better gauge R&R (repeatability and reproducibility), for both pressure decay and mass flow leak testing.
As a rule of thumb, the largest leaks are generally found with air. However, as leaks get smaller, or as the need for speed and certainty increases, manufacturers must invest in more sensitive equipment that uses helium or hydrogen.
Sometimes, more than one method is used. For instance, helium is commonly used for pinpointing leaks after pressure decay methods determine that a leak is occurring.
Each testing method has pros and cons that engineers must carefully consider.
"There is not one method that is the best for all challenges," warns ATC's Davidson. "There are a number of criteria that need to be investigated for every leak testing application, [such as] sensitivity, reliability, flexibility, material makeup, permeation of materials, cost and testing speed. It is always best to consult with experts in this field before choosing a method for your [application]."
“By a considerable margin, pressure decay continues to be the most popular method to test disposable medical devices,” claims David Kralovetz, regional manager at Cincinnati Test Systems Inc. “We are seeing many more wearable devices being tested for IP67 ratings.
“For most applications, pressure decay has far better sensitivity vs. mass flow testing, largely due to the precision of the pressure transducers,” explains Kralovetz. “Pressure decay is also far more repeatable, due to the isolation of test air within the part.”
According to Kralovetz, air, nitrogen and vacuum are the most common media used for testing disposable medical devices. For more durable, implantable devices, helium mass spectrometer testing is frequently used, due to the extremely low leak rates of those devices.
“Performance requirements always dictate the use of helium,” says Jacques Hoffmann, president of InterTech Development Co. “That’s why implantable devices, such as pacemakers and infusion pumps, are typically tested with helium. Dry air is usually used to test invasive devices, such as catheters and surgical instruments.”
“Some medical devices, including catheters, have unique testing requirements,” notes Paul Chamberlain, CEO of LACO Technologies Inc. “This requires that a leak tester be able to have multiple test functions built in, such as pressure decay, occlusion and cracking pressure.
“Air leak testing methods are still the most common type used in the medical device industry,” says Chamberlain. “But, helium tracer gas testing is becoming more [popular for some applications].”
“Medical device manufacturers want leaks tested with a small footprint, simple user interface and good repeatability,” adds Burch. “More and more medical devices are being manufactured by start-up companies that often have limited resources. They prefer to use nitrogen, because it is clean and dry. Nitrogen also allows for a much higher test pressure than a conventional compressor.
“Most of the medical device leak testers we sell are pressure decay instruments,” explains Burch. “Typically, pressure decay leak testers are more tolerant of contamination than mass flow testers and they don’t require a reset in the event of a large leak.”
No matter what type of leak testing method is used, many medical device manufacturers have increasingly tighter leak rate requirements today.
“In some applications, there are tighter leak testing requirements,” says Chamberlain. “Newer products, such as implants and wearable devices, often require tighter leak rates compared to [traditional] disposable medical devices. We are also seeing requirements for higher speed testing to increase leak testing throughput.”
Testing Challenges
When testing medical devices, engineers often encounter numerous challenges. For instance, a major factor in successful leak testing is correct
clamping and sealing of the test piece.
Medical devices frequently have small orifices and multiple transfer paths to test. This can make sealing and clamping more difficult than other leak testing applications.
“Good fixturing is critical for achieving repeatable results,” claims ATEQ’s Burch. “I often must explain to customers and machine builders the need for positive stops on fixtures.
“Any flexible product also presents a challenge with leak testing,” says Burch. “When possible, we always prefer to constrain the device under test while avoiding masking leak paths.”
Flexible components, such as plastic tubing, can create problems because of fluctuations in volume created by the distortion of the material. For instance, with ventilator parts, multiple precise pressures may be required to test each component.
“For small parts, fixturing can be a very critical point for the leak test,” says ATC’s Davidson. “On the other hand, some parts, such as catheters, often come with a standard luer connected, which is very easy to connect to.
"With air leak tests—flow or pressure—flexibility is a tricky point," notes Davidson. "Here, fast stabilization times are needed to ensure a reliable measurement. Especially for pressure-based methods, it also is critical to know the exact volume of the deflated parts to ensure correct conversion from pressure drop into leak rate."
According to InterTech Development’s Hoffmann, expandable devices, such as bags and catheters, are the most difficult products to test. “They cannot be accurately tested with low-cost pressure decay instruments,” he points out.
“Small, flexible materials are always a challenge, because they’re difficult to handle,” adds Inficon’s Hoffman. “We recently developed several new leak testing products that are designed to reduce this time-consuming process”
Earlier this year, Inficon unveiled the Contura S400, which cradles soft medical devices to equalize forces on both the inside and outside of the part. Two elastic membranes tightly enclose the device to be tested in a film chamber by creating a vacuum. The machine analyzes the pressure profile in just seconds without damaging the medical device.
“A soft shell goes around the product being tested to limit the forces across the product as it measures the escaping gas,” says Hoffman. “In the past, some older pressure testing techniques would cause flexible components to explode.”
Industry 4.0 is also changing the way that leak testing equipment is being designed for new medical device applications. One growing trend is data traceability.
"Today, medical device manufacturers want leak testing equipment that does more than just find and eliminate faulty components," notes Zeltwanger's Goehner. "They want to track and trace components [and monitor] quality assurance data, such as leak testing measurements. This data needs to be traceable to the serial number of the product, and it needs to be easily gathered, analyzed and stored."
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!




