New Plastics for Medical Devices
New materials are meeting the varying performance demands of medical devices

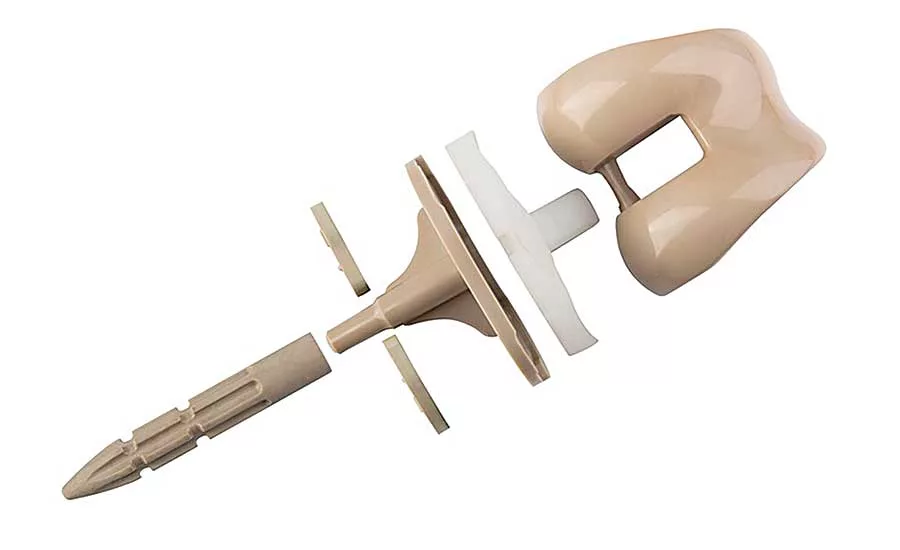
A key aspect of this prosthetic foot is its keel—a single, S-shaped part, molded from Hytrel 8238, that acts like a spring to store and release energy as the person walks. Photo courtesy DuPont
This knee implant features parts molded from fiber-reinforced PEEK. The implant lasts longer and costs less than metallic implants. Photo courtesy Solvay Specialty Polymers

This tray for sterilizing endoscopic instruments is molded from a clear, heat-resistant polycarbonate. Photo courtesy Covestro

Lightweight, high-strength polymers from RTP Co. enabled Plásticos Mueller to develop an innovative wheel-and-gear system for wheelchairs. Photo courtesy Plásticos Mueller
Medical devices represent both a challenge and an opportunity for plastics manufacturers.
On the one hand, medical devices give chemical engineers the opportunity to push the limits of polymer formulation. Disposable or reusable, stiff or flexible, clear or opaque, tiny or massive—there’s a material for any application. There are plastics designed for lubricity and plastics for a good grip. There are plastics formulated with antimicrobial agents, plastics that dispel static electricity, and plastics designed to replace hips, knees and other joints. There are even bioabsorbable polymers for temporary implants.
On the other hand, engineers have to deal with issues that don’t come into play when formulating materials for auto parts or appliances. Medical devices must be cleaned constantly, and they may need to be sterilized. The material might need to be biocompatible, and it will certainly have to pass a slew of regulatory hurdles.
Here’s a look at some of these unique materials.
Strong, But Flexible
DuPont Hytrel is a plasticizer-free thermoplastic polyester elastomer (TPC-ET) for applications requiring maximum flexibility. This resilient material provides excellent flex fatigue resistance and spring-like properties. It can be used over a wide range of temperatures while still retaining its flexibility and mechanical properties. It enables the design of a variety of parts and products that combine the best features of both high-performance rubbers and flexible plastic materials.
Available with hardness ratings ranging from 30 to 82 Shore D, Hytrel can be processed with a variety of molding or extruding technologies.
One company that is taking advantage of this material’s unique properties is Niagara Prosthetic & Orthotics International Corp. of St. Catharines, ON. In 2012, the company introduced the Niagara Foot, an affordable, high-performance prosthetic foot designed for individuals with active lifestyles and those who walk on rugged ground. The foot is designed to mimic the natural movement of the ankle complex. For use by active adults weighing up to 176 pounds, the foot is available in a variety of sizes with an optional cosmetic cover. Prosthetists can easily customize the response of the Niagara Foot to the user’s weight, activity levels and personal preferences.
Looking for quick answers on assembly and manufacturing topics? Try Ask ASM, our new smart AI search tool. Ask ASM
A key aspect of the prosthetic foot is its keel—a single, S-shaped part, molded from Hytrel 8238, that acts like a spring to store and release energy as the person walks.
“The energy-return principle incorporated into the design of the Niagara Foot allows the user to walk more naturally than conventional designs,” explains Rob Gabourie, founder of Niagara Prosthetic & Orthotics and inventor of the Niagara Foot. “Patients are able to detect and appreciate the performance of this device, which decreases the muscular effort required for walking.”
The combination of low flex fatigue and high stress resistance provided by Hytrel was critical to achieving this concept. Indeed, Gabourie is convinced that his design will not work with any other material. Whereas other materials, including polyoxymethylene (POM) and polyamides, were unable to pass 2 million cyclic loads due to deformation or, in some cases, structural failure, Hytrel exceeded that milestone during testing according to ISO 10328 standards.
“Of particular interest to Niagara is the ability of Hytrel to store and release energy,” states Dan Curran-Blaney, application development representative at DuPont Canada. “The material must also be able to endure severe cycling of loads, as experienced in normal usage. The unique attribute of Hytrel is its ability to recover its shape, while continuously cycling under load, which enhances its spring-like properties.”
Hytrel’s ease of processing also facilitated manufacture of the prosthetic foot. Voids were prevalent in parts molded with POM or polyamide, but were absent from parts molded from Hytrel.
“The geometry of the current design requires abnormally thick wall sections,” explains Helga Plishka, senior technical representative at DuPont Canada. “Because Hytrel flows easily to produce a well-packed part, the occurrence of voids…was eliminated. That was essential for achieving flex fatigue resistance.”
PEEK Performance
One of the newer thermoplastics for medical devices is polyetheretherketone (PEEK). This material is highly resistant to thermal degradation as well as organic and aqueous environments. PEEK has outstanding wear resistance, good dielectric strength, volume resistivity, tracking resistance and excellent radiation resistance.
These properties make the material ideal for long-term orthopedic implant applications, such as bone screws, plates and pins, tissue anchors and suture screws. It’s also being used to replace metal as a hip stem component.
Last November, Solvay Specialty Polymers introduced Zeniva ZA-600 CF30 PEEK, a 30 percent carbon fiber-reinforced, radiolucent polymer for implantable device applications. The material has a modulus similar to cortical bone. That means, unlike implantable metals, it can help implants minimize reduction in bone density by maintaining normal stress on surrounding bone tissue.
Zeniva ZA-600 CF30 is twice as strong as unmodified PEEK, making it a candidate for structural, load-bearing implantable devices used in spine, hip and knee replacements. This quality allows designers to reduce the size and scale of implanted devices to make them less intrusive. Like unmodified PEEK, Zeniva ZA-600 CF30 resists creep and withstands prolonged fatigue strain.
And, like unmodified PEEK, Solvay’s new polymer is inherently radiolucent, giving it an advantage over metals, which prohibit visualization of implants and fusions using X-ray, MRI and other medical imaging methods.
In March, Okani Medical Technology, a Chinese manufacturer of artificial joints, introduced an all-polymer knee implant based on Zeniva PEEK. Okani’s ORGKnee implant offers a longer service life at a lower cost than traditional metallic implant systems.
Okani evaluated suitability of Zeniva PEEK for the femoral and tibial tray components of its knee implant according to ISO 14243-1:2009 test method, which simulates normal walking behavior over 10 years. The findings indicate that the ORGKnee implant exhibited 50 percent less wear vs. a metal implant as measured by material loss over 3 million cycles.
“PEEK’s injection molding capability makes large-scale production of ORGKnee implants possible in a fraction of the time and cost it takes to make metal implants, which can take up to three months to manufacture, machine and polish using methods that can pose risks both to workers and the environment,” adds Zhonglin Zhu, chief technology officer for Okani.
The ORGKnee implant entered pre-clinical trials in April, and will undergo standard clinical trials by China’s Food and Drug Administration in September. If the trials are successful, Okani plans to commercially launch ORGKnee in 2020.
Polycarbonate Takes the Heat
Polycarbonate is one of the most popular plastics for medical devices. The material resists impacts, UV light and high heat. It is clear, dimensionally stable and flame retardant. It withstands sterilization with either gamma rays or ethylene oxide, and it can be assembled with ultrasonic welding. Typical applications include reservoirs, high-pressure syringes, artery cannulas, stopcocks, Luer-Lok fittings, centrifugal force separators, blood filter housings and dialyzer housings.
When Tristel Solutions was looking to design a faster, better system to clean and disinfect endoscopic and ultrasound instruments, polycarbonate was the natural way to go. For its Stella cleaning system, Tristel required a biocompatible, heat-resistant and medical-grade polycarbonate that could withstand autoclave temperatures as high as 134 C.
Apec 1745 high-heat polycarbonate from Covestro fit the bill. This transparent and strong co-polycarbonate is suitable for medical applications requiring autoclave sterilization and features additional benefits like easy release, high softening temperature, good hydrolysis resistance and biocompatibility according to many ISO 10993-1 test requirements. Applications include baby incubators, dental or surgical instruments, sterilization boxes and endoscope disinfectant systems.
The material’s glass-like transparency was important to Tristel Solutions, since users of the Stella system need to monitor the disinfection process. Easy processing of the material allows the large contamination tray to be molded in one piece, which reduces assembly time and eliminates the risk of liquid leakage.
High-Strength Gears
A good wheelchair can be of tremendous benefit to a person otherwise unable to get around due to illness or injury. Most of us don’t realize just how much pain and risk can be involved in their day-to-day use.
“Wheelchair propulsion is a highly complex movement that requires short, fast, continuous effort with both hands and both arms; the stress on the shoulders and wrists can be significant, and can cause people intense pain,” explains Paulo Rodi, research and innovation manager with Plásticos Mueller, an injection molding company in Sao Paulo, Brazil.
That stress is greatly intensified if the user tries to climb a slope. There’s also a very real danger of rolling backwards.
In 2009, Mueller saw an opportunity to significantly improve wheelchair design with a new type of wheel system that, like a multispeed bicycle, could use a gear system to make hill climbing easier. In addition, the wheel would automatically brake if the chair began to roll backwards.
“Our calculations suggested that we could increase safety and reduce the torque needed to move the chair by upwards of 50 percent,” explains Rodi. “This would greatly improve the mobility and quality of life for many people.”
But the Mueller team’s mission quickly reached an impasse: The gears needed to stand up to extreme friction and stress. The wheels would have to be very strong, too, but they couldn’t weigh any more than the conventional wheels they planned to replace.
Some lightweight, high-strength polymers from RTP Co. solved the problem. For the gears, RTP supplied its 4000 series glass-fiber-reinforced polyphthalamide with Teflon lubrication. For the wheels, RTP supplied 100 series polypropylene reinforced with 30 percent very long glass fibers.
The result was dubbed the Mueller easyMOB system, a geared 24-inch wheel designed to be fitted to most commercial wheelchairs.
Mueller established an extensive array of validation tests for the wheel and gear system, and the prototypes made with RTP compounds passed them all with flying colors.
“We ran the wheel on a dynamometer under heavy load at the typical 200,000 cycles, and were amazed at how little wear there was,” says Rodi. “So we ran it up to 1 million cycles, and it still performed brilliantly.”
Wheelchair users have been delighted with the new easyMOB wheels, and Mueller is seeing high levels of interest in the system from wheelchair manufacturers around the world. Mueller is now planning to create an expanded line of wheels using the same compounds and know-how they developed with RTP.
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!