Medical Device Manufacturer Uses Adhesive to Encapsulate Sensors for Prosthetic Limbs
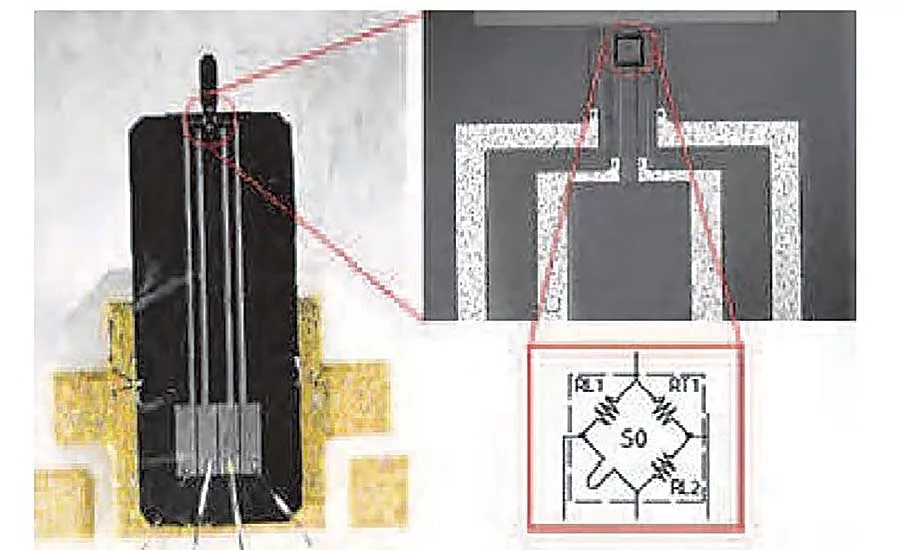
Implantable orthopedic devices and the wireless sensors that monitor them are highly specialized products that regularly come into contact with body tissues and fluids. Because of this, each internal part in the product, including adhesive, is required by the FDA to be biocompatible with living tissue to prevent human toxicity.
Recently, University of Tennessee researchers tested four medical-grade adhesives to determine their biocompatibility and reliability levels as an encapsulant of tiny microcantilever-based sensors within a prosthetic for the upper tibia. The sensors accurately measure how a repaired knee joint performs following total replacement (arthroplasty) surgery.
Arthroplasty alleviates arthritic pain by capping the ends of the bones that form the joint and kneecap with metal and plastic. An invasive surgery, the procedure resurfaces the damaged segments of the knee joint, and is generally performed following unsuccessful attempts to treat the existing bones. When successful, arthroplasty lessens osteoarthritis (a degenerative joint disease), rheumatoid arthritis (pain due to inflammation) and traumatic arthritis (due to injury).
In this research project, the researchers installed 30 microcantilever-based sensors in the prosthetic to create an accurate, yet computationally efficient, data map. They also intentionally chose non-cost-prohibitive sensors to mimic the market forces that medical professionals would face when selecting sensors for a similar application.
The researchers decided to encapsulate the sensors to protect them from damage and to form an appropriate interface between the implanted device and the bone tissue. All of the sensors, along with a force sensitive resistor, were encapsulated in 2 millimeters of epoxy. A moderate force was then applied to both for several minutes.
Of the four epoxies tested, EP30Med from Master Bond Inc. performed the best. Researchers concluded that this was due to the two-component adhesive’s low viscosity, non-rapid cure time, and minimal amount of air bubbles present during the curing process. Together, these factors produced a homogenous material and a durable bond. Equally important, the adhesive is biocompatible, per USP Class VI testing.
On the downside, however, the researchers did note some epoxy shrinkage and that encapsulating such a large number of sensors is labor intensive and time-consuming. Nevertheless, they concluded that EP30Med is a sound choice for such a procedure.
Looking for quick answers on assembly and manufacturing topics? Try Ask ASM, our new smart AI search tool. Ask ASM
Since this project, the adhesive has also been used to encapsulate microcantilever sensors in high-frequency resonators and photothermic spectroscopes. Other uses include high-performance bonding, sealing and coating applications in food production and safety, as the adhesive meets FDA CFR 175.105 and 175.300.
EP30Med has a four-to-one mix ratio by weight and contains no solvents or diluents. It is optically clear; bonds well to metal, plastic, glass, wood and ceramic; resists water, oils and many organic solvents; and is serviceable over a temperature range of -60 to 250 F. To optimize performance, the recommended cure schedule is overnight at room temperature.
Key non-medical industrial certifications of the adhesive include MIL-STD-810G for fungus resistance and MIL-STD-883J for thermal stability. Packaging options range from cans and pails, to standard syringes, premixed and frozen syringes, and the FlexiPak divider pouch.
For more information on medical-grade adhesives, call 201-343-8983 or visit www.masterbond.com.
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!