Vision System Performs Flawless Blister-Pack Inspection

The Lynx Spectra HR vision system is designed to inspect up to 300 blister packs per minute. Photo courtesy Stemmer Imaging
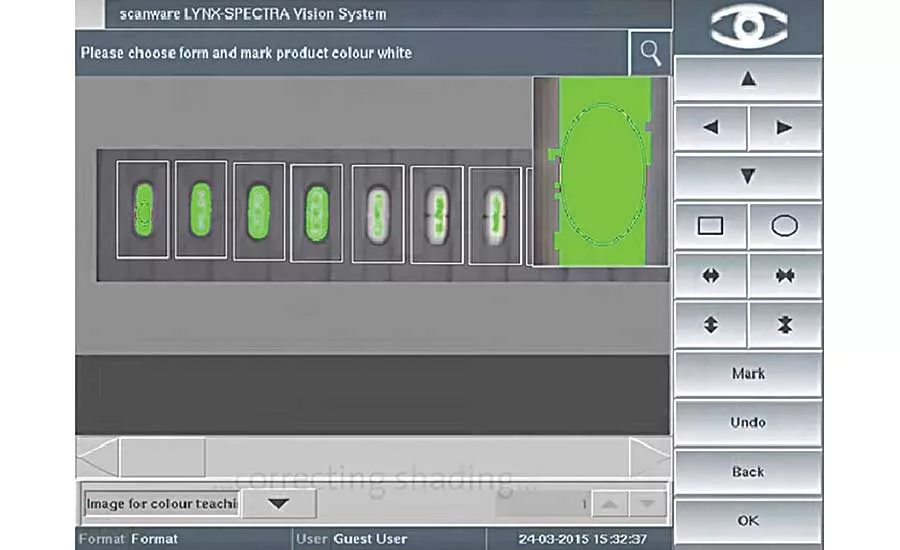
Various product-related tolerances (such as color and shape variations) can be programmed for the same blister. Photo courtesy Stemmer Imaging
Pharmaceutical OEMs and their suppliers know all about the concept of ‘Japan quality’ products. This phrase refers to packaged pharmaceuticals that are flawless in appearance. For, in the Japanese culture, perfect packages indicate a strong likelihood of perfect contents (tablets and capsules). To meet this difficult challenge, small and large manufacturers rely on machine vision systems to perform accurate and consistent inspection of these medications sealed in blister packs.
Scanware Electronic GmbH, based in Bickenbach, Germany, has been designing and building custom vision systems since 1989. Managing Director Harald Mätzig says the company has installed more than 2,000 systems worldwide in good manufacturing practice (GMP) facilities operated by pharmaceutical manufacturers. Each system is highly specialized and designed for these facilities, which require strict environmental control to assure the manufacture of sterile, potent and uncontaminated products for human therapies or consumption.
In fall 2015, Scanware developed the Lynx Spectra HR system in close cooperation with Stemmer Imaging, a Puch-heim, Germany-based company that specializes in system design consultancy, and delivering and supporting various system components. For this project, SI provided six 3CCD CV-M9 CL color cameras from Danish supplier JAI.
Mätzig says developing this vision system presented two major challenges for the collaborators. The first is inspecting up to 300 blister packs—with ever-so-slightly different configurations—per minute. Because each pack has three rows, that means the system has to accurately inspect 900 blisters per minute.
Challenge two is attaining Japan quality for each and every blister. Rejection parameters include defects or any particles on the tablets or the coating, extraneous particles and contamination with hair. Particles may not exceed 0.3 millimeter and must be detected with extremely high color differentiation.
The system lets operators program various product-related tolerances (such as color and shape variations) for the same blister. It also identifies and localizes error clusters to eliminate error causes.
Dirk Schneider, head of development at Scanware, says the collaborators decided to use multiple cameras in the system because the mechanical and electrical environmental conditions in and around packaging machines are not conducive to the use of a single high-resolution camera with a large working distance.
Looking for quick answers on assembly and manufacturing topics? Try Ask ASM, our new smart AI search tool. Ask ASM
Depending on the application, two, three, four or six cameras are installed in a cascade arrangement in a small space under a closed stainless steel hood. A water-cooling system offsets heat generated by the cameras.
Each camera features three 0.33-inch scan CCD sensors that supply 30 images at a resolution of 1,024 by 768 pixels (pixel size is 4.65 microns). During operation, the lens breaks entering light into its red, green and blue components and guides them to each respective sensor. This action results in precise color separation with limited variation of brightness or color.
Image information from all cameras is combined by a multiplexer (developed by Scanware) and prepared for evaluation. As a result, the operator sees only a combined image for evaluation.
Another key feature of the vision system is a white light LED module, which operates in flash mode with a frequency of about one millisecond. System operators reduce the module’s illumination on bright products and increase it for dark ones.
For more information on CCD-sensor cameras, call 800-445-5444 or visit www.jai.com/en.
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!



